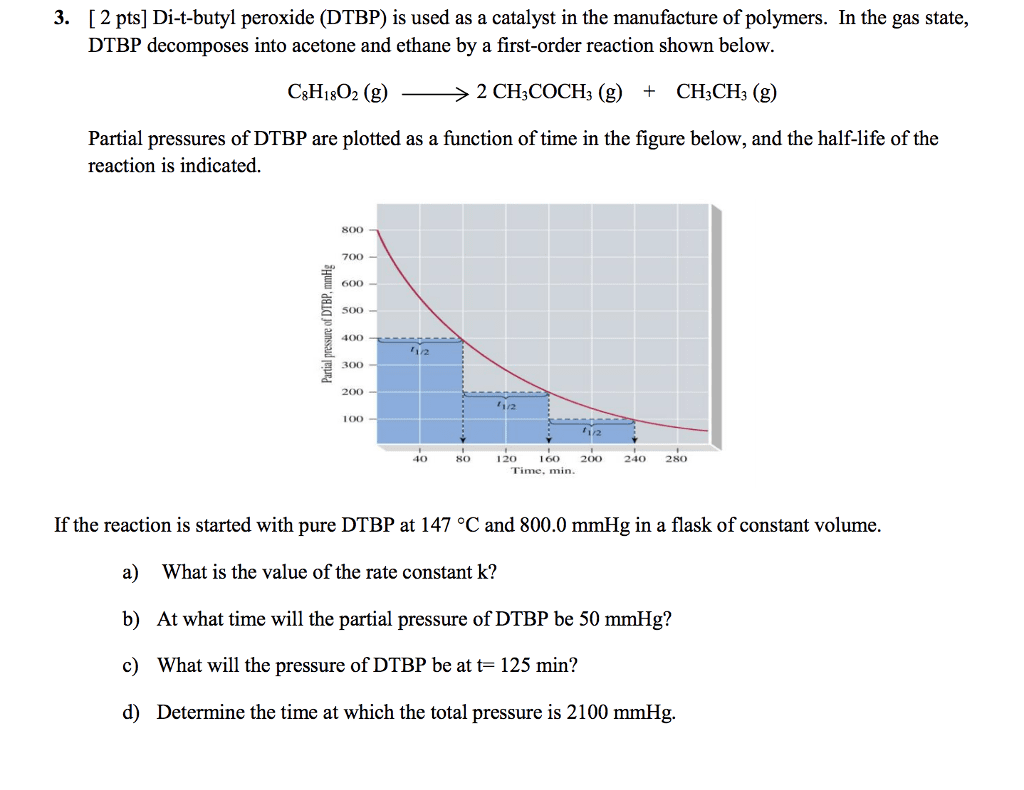
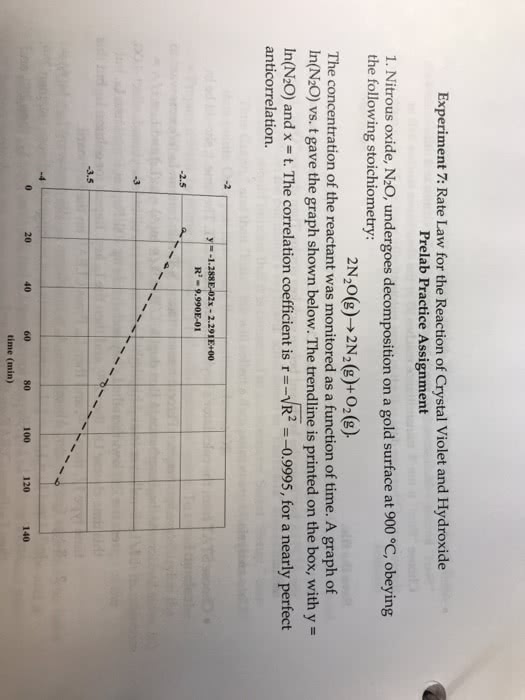
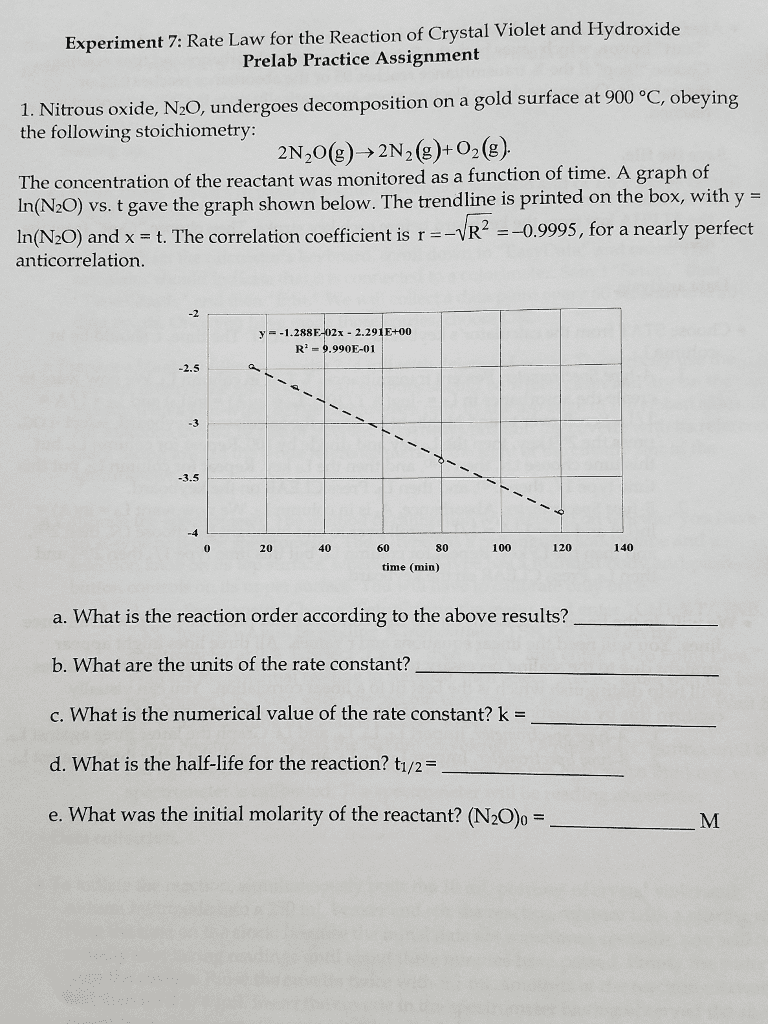
1
answer
0
watching
278
views
16 Jun 2020
The decomposition of ethylene oxide, (CH2)2O(g) → CH4 (g) + CO (g), at 690K is monitored by measuring the total gas pressure as a function of time. The data obtained are listed in the table below.
Time (min)
Ptotal (mmHg)
10
139.14
20
151.67
40
172.65
60
189.15
100
212.34
200
238.66
∞
249.88
What is the rate constant?
The decomposition of ethylene oxide, (CH2)2O(g) → CH4 (g) + CO (g), at 690K is monitored by measuring the total gas pressure as a function of time. The data obtained are listed in the table below.
| Time (min) | Ptotal (mmHg) |
| 10 | 139.14 |
| 20 | 151.67 |
| 40 | 172.65 |
| 60 | 189.15 |
| 100 | 212.34 |
| 200 | 238.66 |
| ∞ | 249.88 |
What is the rate constant?
1 Sep 2020