1
answer
0
watching
772
views
8 Jul 2020
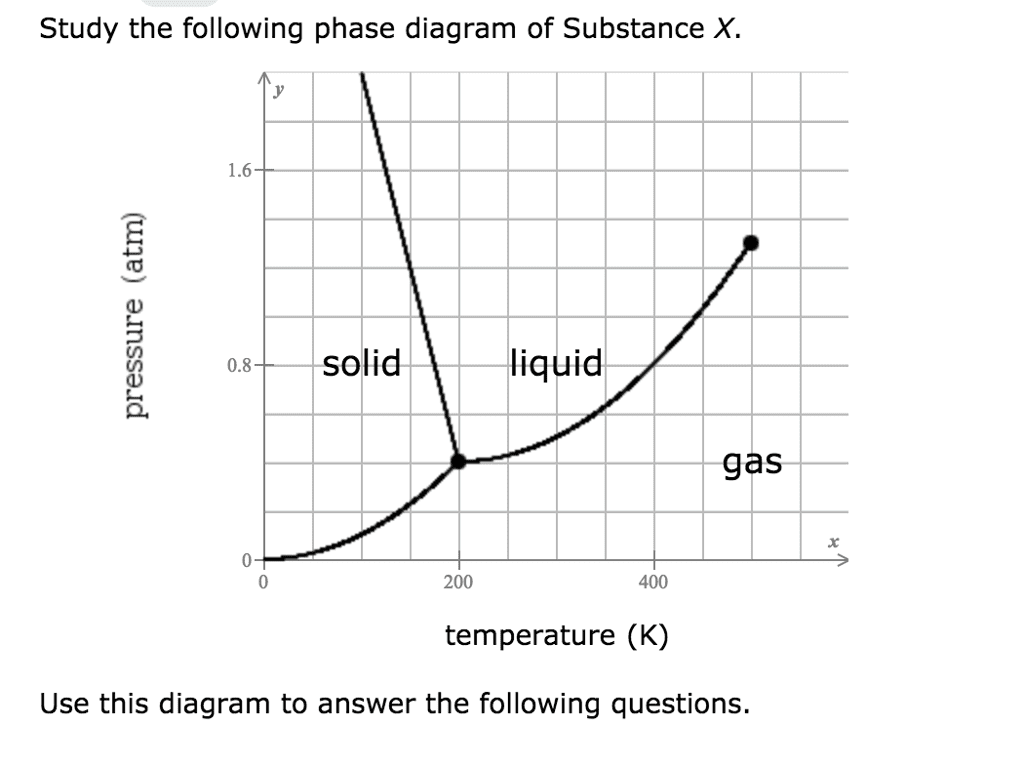
On the following typical phase diagram, which statement is CORRECT?

On the following typical phase diagram, which statement is CORRECT?

1
answer
0
watching
772
views
For unlimited access to Homework Help, a Homework+ subscription is required.
24 Jul 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Points A to G are located on the phase diagram of water. Which of the following statements are correct regarding navigation from one point to another across the phase diagram?
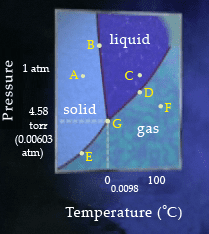
CHECK ALL THAT APPLY.
Check all that apply.
| Moving from point A to point C, the temperature increases. | |
| To move from point D to point F, you must decrease both the temperature and the pressure. | |
| At point E, the temperature is less than 0 degree celsius%u2218C | |
| To move from the point G to point F, you must increase both the temperature and the pressure. | |
| To move from point C to point D, you must decrease only the pressure. | |
| To move from point G to point B, you must increase the temperature. |
Points A to G are located on the phase diagram of water. Which of the following statements are correct regarding navigation from one point to another across the phase diagram?