3
answers
5
watching
932
views
6 Oct 2020
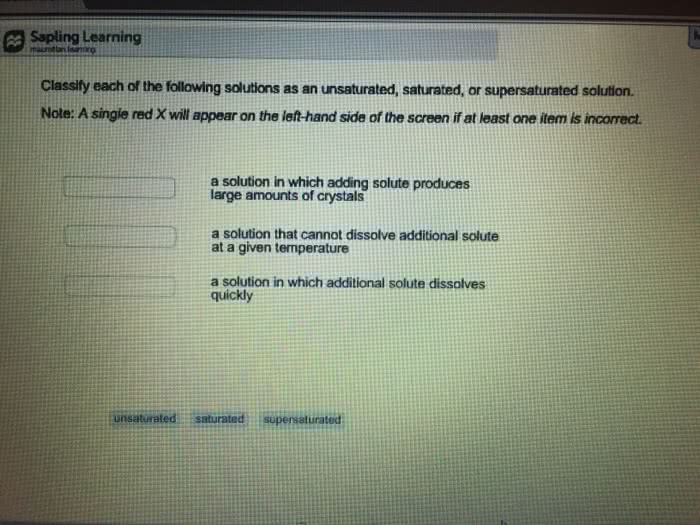
Classify each of the following solutions as saturated, unsaturated, or supersaturated based on the following observations:
(a) Agitation of the solution produces a large amount of solid crystals.
(b) Heating the solution causes excess undissolved solute present to dissolve.
(c) Excess undissolved solute is present at the bottom of the solution container.
(d) The amount of solute dissolved is less than the maximum amount that could dissolve under the conditions at which the solution exists.
Classify each of the following solutions as saturated, unsaturated, or supersaturated based on the following observations:
(a) Agitation of the solution produces a large amount of solid crystals.
(b) Heating the solution causes excess undissolved solute present to dissolve.
(c) Excess undissolved solute is present at the bottom of the solution container.
(d) The amount of solute dissolved is less than the maximum amount that could dissolve under the conditions at which the solution exists.
3
answers
5
watching
932
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Read by 2 people
5 Apr 2022
Read by 2 people
25 Mar 2022
Already have an account? Log in
Neal BroomheadLv7
15 Nov 2020
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232