1
answer
0
watching
276
views
28 Nov 2020
1) The conjugate acid of 
A. 
B. 
C. 
D. 
2) The conjugate base of 
A. 
B. 
C. 
D. 
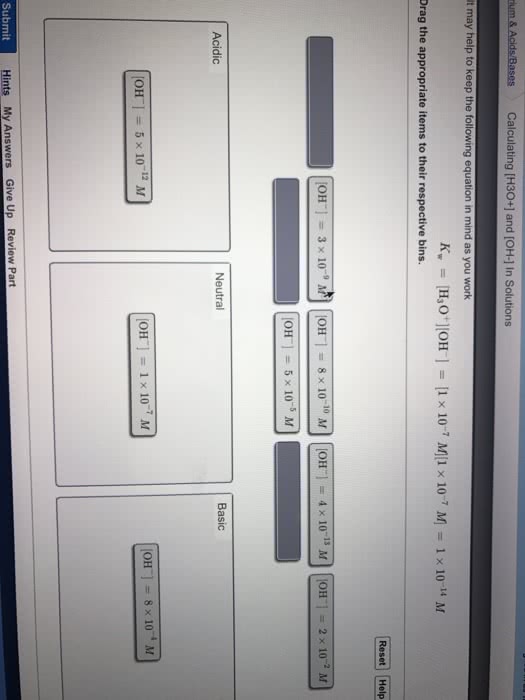
3) In an acidic solution, how does the concentration of hydronium ions compare to the concentration of hydroxide ions?
A. [Hydronium ion] is greater than [hydroxide ion].
B. There is not enough information to tell.
C. They are equal.
D. [Hydronium ion] is less than [hydroxide ion].
1) The conjugate acid of 
A. 
B. 
C. 
D. 
2) The conjugate base of 
A. 
B. 
C. 
D. 
3) In an acidic solution, how does the concentration of hydronium ions compare to the concentration of hydroxide ions?
A. [Hydronium ion] is greater than [hydroxide ion].
B. There is not enough information to tell.
C. They are equal.
D. [Hydronium ion] is less than [hydroxide ion].
Robert KubaraLv10
16 Jan 2021