4
answers
0
watching
85
views
9 Jan 2021
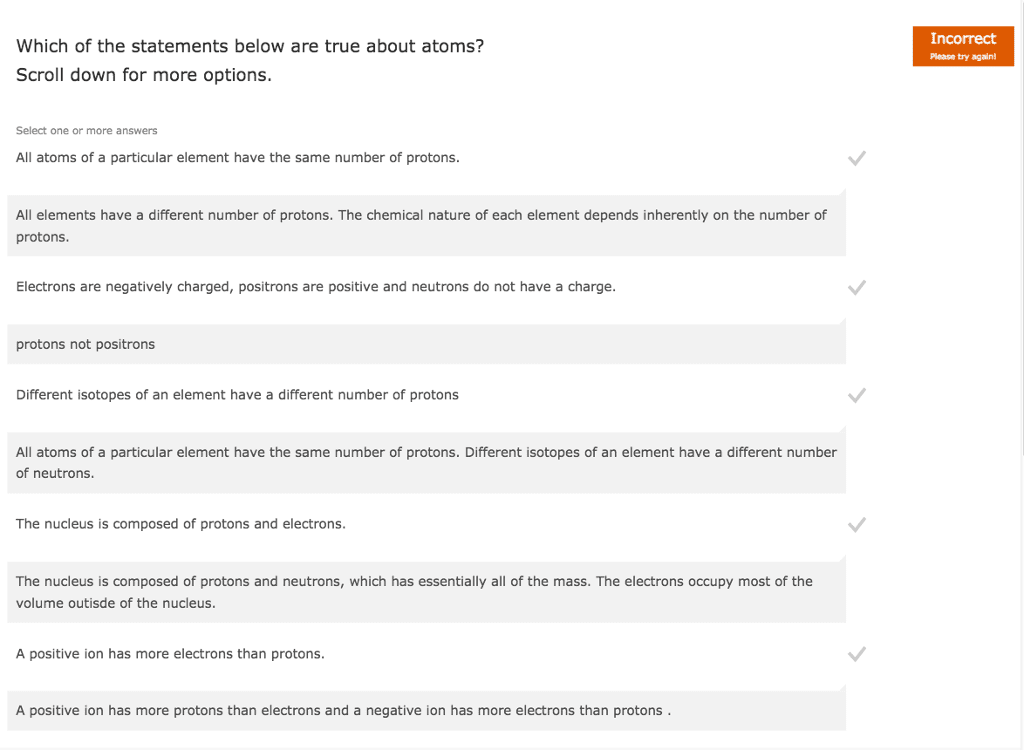
Which of the following statement/s is/are true?
I- The masses of the proton and electron are essentially the same which only differs in the fourth significant figures.
II- The atomic number of an element is the same as the number of neutrons in the element.
III- The mass number of a neutral element is equal to the sum of the numbers of electrons and neutrons of that element.
Which of the following statement/s is/are true?
I- The masses of the proton and electron are essentially the same which only differs in the fourth significant figures.
II- The atomic number of an element is the same as the number of neutrons in the element.
III- The mass number of a neutral element is equal to the sum of the numbers of electrons and neutrons of that element.
4
answers
0
watching
85
views
For unlimited access to Homework Help, a Homework+ subscription is required.
papayaprofessorLv10
5 Sep 2022
Already have an account? Log in
mayankjalan96Lv10
3 Sep 2022
Already have an account? Log in
John Edward CayasLv10
9 Jan 2021
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232