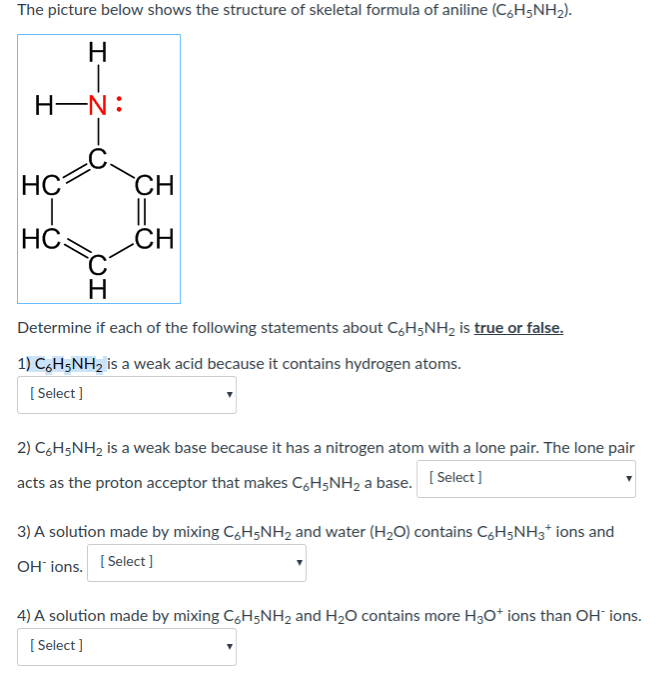
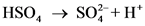One acid–base classification defines a base as a substance that acts as a proton (H+H+) acceptor, and is also known as a Bronsted–Lowry base. All bases contain a non‑bonding pair of electrons.
In each of the molecules, identify the atom that behaves only like a Bronsted–Lowry base.







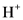 (hydrogen ion) or proton. In general Bronsted-Lowry acid is a proton donor.
(hydrogen ion) or proton. In general Bronsted-Lowry acid is a proton donor.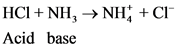
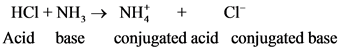
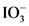 is the conjugate base of
is the conjugate base of 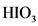 .
.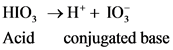
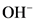 is the conjugate acid of
is the conjugate acid of 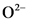 .
.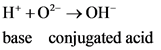
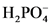 will from after accept one proton by
will from after accept one proton by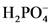 as follows:
as follows: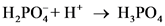
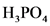
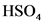 will from after donate one proton from
will from after donate one proton from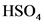 as follows:
as follows: