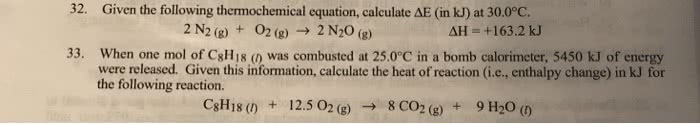2
answers
0
watching
37
views
23 Nov 2019
How much heat is released when 75 g of octane is burned completelyif the enthalpy of combustion is -5500 kJ/mol C8H18?
C8H18 + 25/2 O2 --> 8CO2 + 9H2O
*****Please show all work and give answer
How much heat is released when 75 g of octane is burned completelyif the enthalpy of combustion is -5500 kJ/mol C8H18?
C8H18 + 25/2 O2 --> 8CO2 + 9H2O
*****Please show all work and give answer
20 Mar 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Collen VonLv2
3 Oct 2019
Get unlimited access
Already have an account? Log in