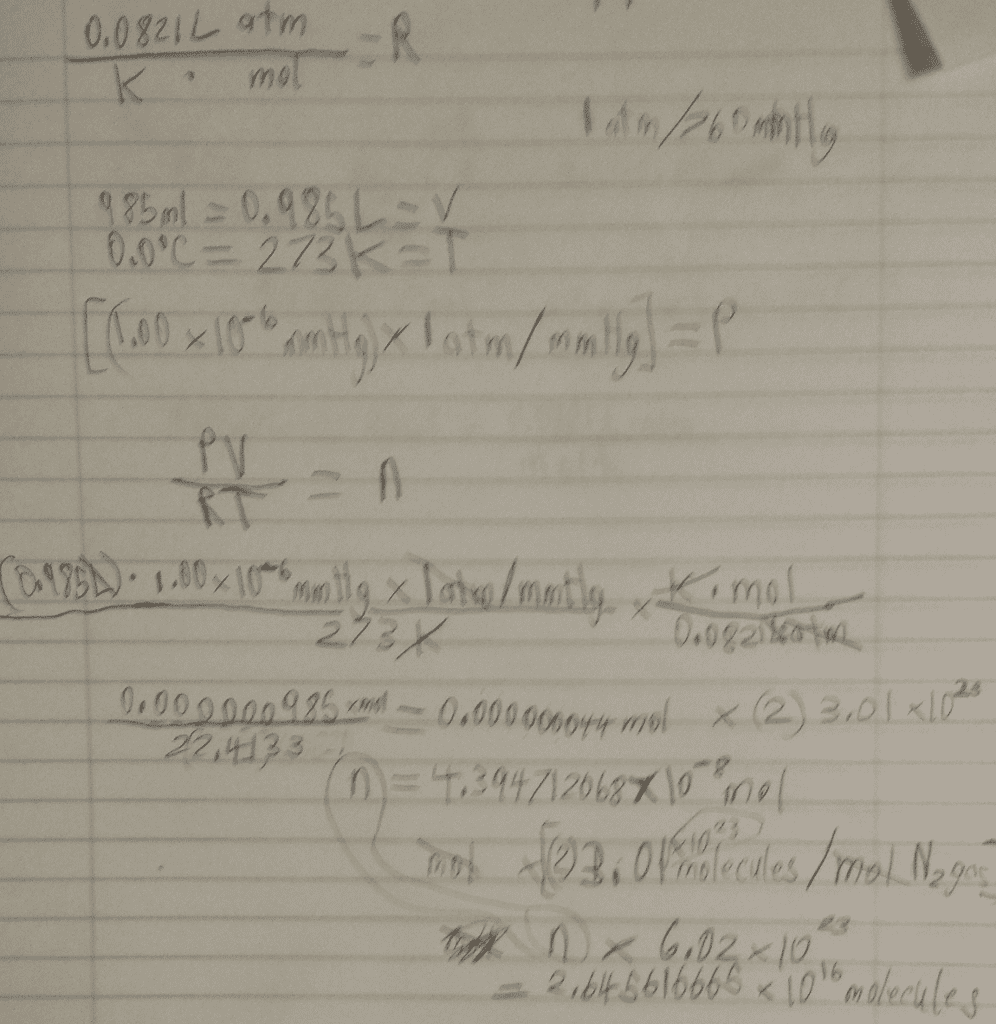4
answers
0
watching
29
views
11 Dec 2019
2) How many molecules of N2 are in a 400.0 mL container at 780 mm Hg and 135°C? Avogadroâs number = 6.022 x 1023
A) 7.01 Ã 1021 molecules
B) 7.38 Ã 1021 molecules
C) 2.12 Ã 1022 molecules
D) 2.23 Ã 1022 molecules
2) How many molecules of N2 are in a 400.0 mL container at 780 mm Hg and 135°C? Avogadroâs number = 6.022 x 1023
A) 7.01 Ã 1021 molecules
B) 7.38 Ã 1021 molecules
C) 2.12 Ã 1022 molecules
D) 2.23 Ã 1022 molecules
30 Dec 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
hasanafat36Lv3
29 Dec 2022
Get unlimited access
Already have an account? Log in
m7mdeljokerLv10
29 Dec 2022
Get unlimited access
Already have an account? Log in
Casey DurganLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in