Biochemistry 2280A Lecture : Topic 9
Document Summary
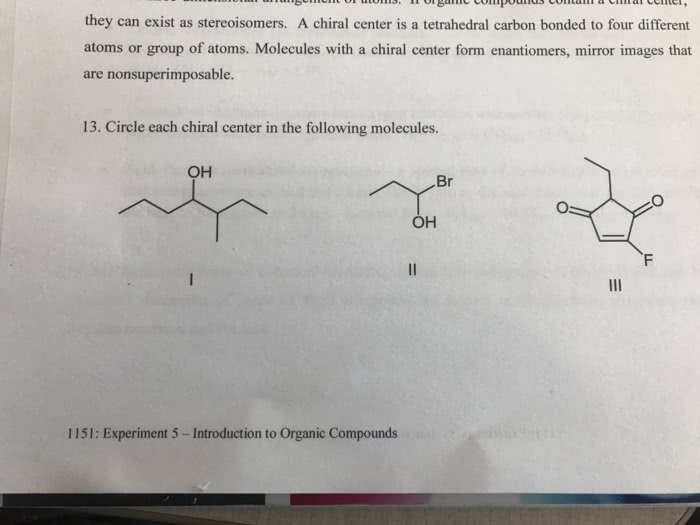
Diagram shown above mirror imagines same 4 atoms bonding but bonded differently cant superimpose them not identical molecules (different orientation in space) enantiomers. Need a naming system to distinguish between each enantiomer different chemically one of the differing properties is that it bends light in different ways plane of polarized light. Guessed and assigned to d to one and l to the other. When the oh is on the right it is d and vice versa. Each has 4 chiral centres this is when fischer projections come in handy. These are all isomers same number of atoms. To number them you start at the c with the carbonyl group. Fructose 1 is the first c on the top next to the carbonyl. Carbon number 5 is d in all of these structures last chiral carbon. These don t stay as straight chain molecules they cyclise. This is in equilibrium as the reverse occurs.