3
answers
2
watching
142
views
6 Oct 2022
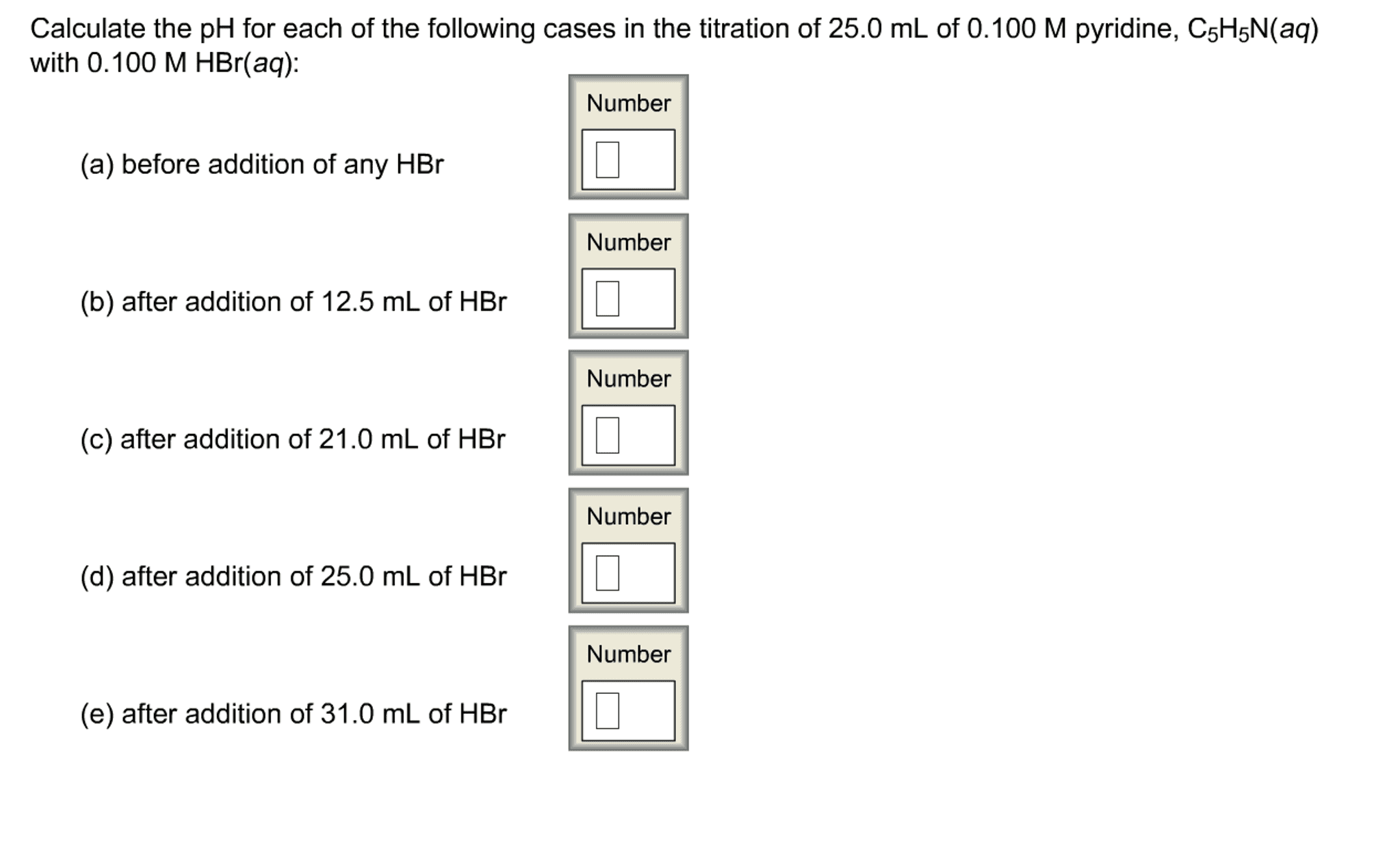
Calculate the pH for each of the cases in the titration of 25.0 mL of 0.200 M pyridine, C5HN(aq) with 0.200 M HBr(aq). The 𝐾b of pyridine is 1.7×10−9.
Before addition of any HBr, after addition of 12.5 mL of HBr, after addition of 18.0 mL of HBr, after addition of 25.0 mL of HBr HBr, after addition of 30.0 mL of HBr HBr
Some explanation of the reasoning for the setup of the first two steps would be appreciated. It is otherwise useless to me. Thank you
Calculate the pH for each of the cases in the titration of 25.0 mL of 0.200 M pyridine, C5HN(aq) with 0.200 M HBr(aq). The 𝐾b of pyridine is 1.7×10−9.
Before addition of any HBr, after addition of 12.5 mL of HBr, after addition of 18.0 mL of HBr, after addition of 25.0 mL of HBr HBr, after addition of 30.0 mL of HBr HBr
Some explanation of the reasoning for the setup of the first two steps would be appreciated. It is otherwise useless to me. Thank you
wahabmunir796Lv10
15 Jul 2023
christopherc63Lv10
10 Nov 2022
Already have an account? Log in
Read by 1 person