1
answer
1
watching
113
views
cmall98Lv1
15 Jun 2023
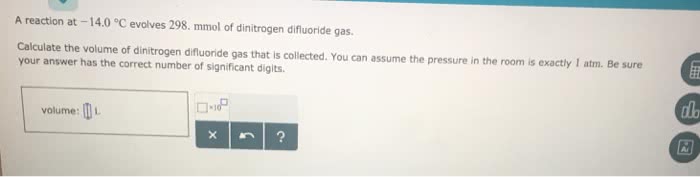
A reaction at −2.0°C evolves 893. mmol of sulfur tetrafluoride gas.
Calculate the volume of sulfur tetrafluoride gas that is collected. You can assume the pressure in the room is exactly 1 atm.
Round your answer to 3 significant digits.
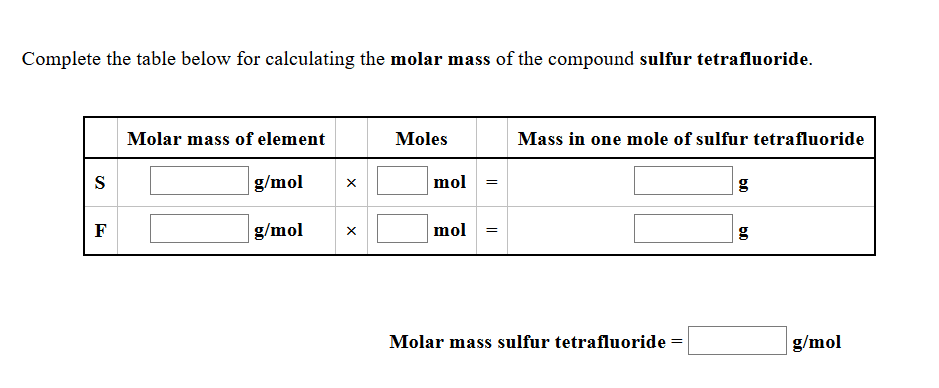
A reaction at −2.0°C evolves 893. mmol of sulfur tetrafluoride gas.
Calculate the volume of sulfur tetrafluoride gas that is collected. You can assume the pressure in the room is exactly 1 atm.
Round your answer to 3 significant digits.
Liked by cmall98 and 1 others
15 Jun 2023