CHEM 222 Study Guide - Separatory Funnel, Diethyl Ether, Gallic Acid
Document Summary
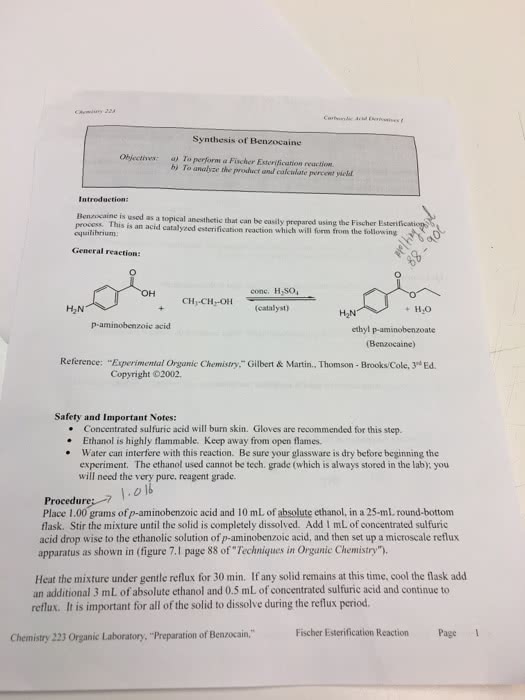
Fischer esterification lab part 1: objective: 1 mark. The objective of this lab is to directly produce the ester, ethyl gallate, by the reaction of a carboxylic acid called gallic acid with ethanol, an alcohol, while using sulfuric acid, as the catalyst. Following the synthesis and isolation of ethyl gallate, characterize the ethyl gallate. Tlc (thin layer chromotography), ir spectroscopy, nmr, and the melting point to verify if it is actually ethyl gallate that is produced: introduction: 2 marks. Brief description of the concept/reaction studied esterification. Fischer esterification is an acid catalyzed process that involves refluxing carboxylic acid with an alcohol. The reflux involves heating the reaction mixture at its boiling temperature and most importantly no compounds are lost when doing this since the condenser catches and returns the escaping vapor (oxford dictionaries, 2012). This technique is used to provide that extra energy to the reaction.