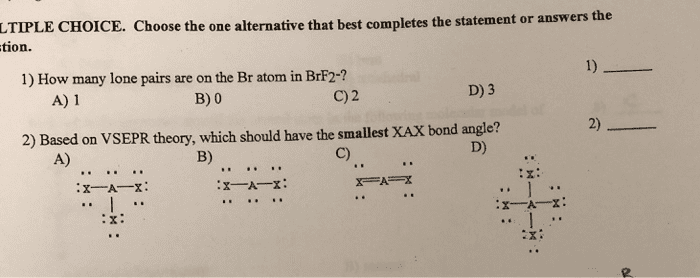LTIPLE CHOICE. Choose the one alternative that best completes the statement or answers th tion. 1) 1) How many lone pairs are on the Br atom in BrF2-? D) 3 A) 1 B) 0 C) 2 2) 2) Based on VSEPR theory, which should have the smallest XAX bond angle? D) A) B) C)
5) Arrange in order from the smallest to the largest bond angle: CH3+, NF3, NH4+, XeF4. 5) A) XeF4, NH4+, NF3, CH3+ C) NF3, NH4+, XeF4, CH3+ B) XeF4, NF3, NH4+, CH3+ D) CH3+, NF3, NH4+, XeF4
11) What is the bond angle in the following molecular model of NO2-? A) less than 109.5 B) 109.5 C) less than 120° but greater than 109.5 D) 120° 12) 12) What are the bond angles in the folowing molecular model of SO32-? ao A) less than 109.5 B) 109.5° C) less than 120° but greater than 109.5 D) 120 13) 13) How many of the following molecules are polar? B) 0 D) 3 E) 1 A) 2 14 14) Determine the electron geometry (eg, molecular geometry (mg. and polarity of Pa A) eg-trigonal pyramidal mg-trigonal pyramidal polar B) eg-tetrahedral mg-bent, polar C) eg-tetrahedral, mg-trigonal pyramidal polar D) eg-linear, mg-linear, nompolar E) eg-trigonal planar, mg-trigonal planar, nonpolar 15) Which orbital hybridization is associated with a tetrahedral charge cloud arrangement? 15)__ B) sp3 D) None of these C) sp 16) Which of the following is not true? 16 A) An sp3 hybrid orbital may hold a lone pair of electrons B) An sp3 hybrid orbital may form a pi bond by overlap with an orbital on another atom. C) An sp3 hybrid orbital may form a sigma bond by overlap with an orbital on another atom. D) The sp3 hybrid orbitals are degenerate.
chlo 17) What orbital hybridization is expected for the central atom in a molecule with a trigonal 17)_ planar geometry? A) sp2 C) sp B) sp3 D) None of these 18) Which atomic orbitals are involved in bonding and which as lone pair orbitals for 18) N2H2? H N N-H A) boading: s on H, sp2 and p on N B) bonding: s on H, sp2 on N C) bonding: sp2 on both H and N D) bonding: s on H, p on N lone pair: sp2 on N lone pair: p on N lone pair: p on N lone pair: sp2 on N 19) Place the following in order of increasing dipole moment 20) Give the hybridization for the O in OF2 21) Give the hybridization for the Br in BrCl3 22) Give the hybridization for the C in HCCH A)sp 20) E) sp3 Ð)sp3d 21) D) sp E)sp3 A)sp3 B) sp3d2 C) sp 22) D) sp3d E)sp2
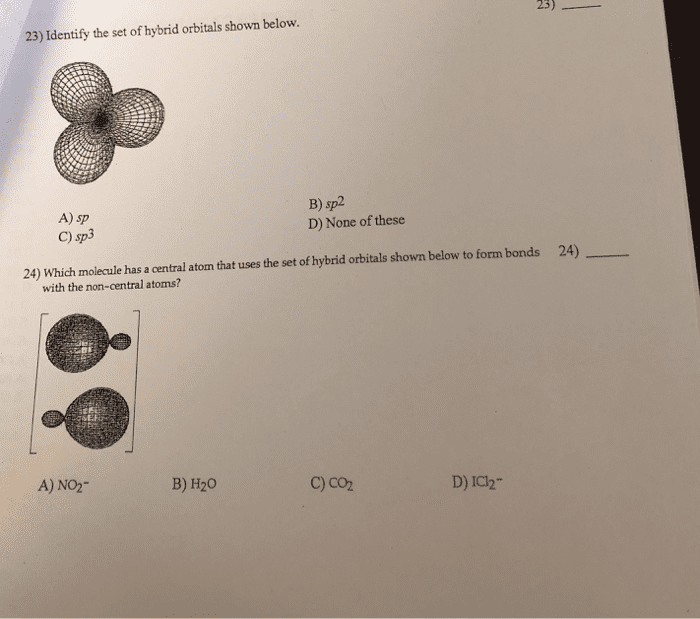
23) Identify the set of hybrid orbitals shown below. A) sp C) sp3 B) sp2 D) None of these 24) Which molecule has a central atom that uses the set of hybrid orbitals shown below to form bonds 24) with the non-central atoms? A) NO2 B) H20 C) co2
Show transcribed image text LTIPLE CHOICE. Choose the one alternative that best completes the statement or answers th tion. 1) 1) How many lone pairs are on the Br atom in BrF2-? D) 3 A) 1 B) 0 C) 2 2) 2) Based on VSEPR theory, which should have the smallest XAX bond angle? D) A) B) C)
5) Arrange in order from the smallest to the largest bond angle: CH3+, NF3, NH4+, XeF4. 5) A) XeF4, NH4+, NF3, CH3+ C) NF3, NH4+, XeF4, CH3+ B) XeF4, NF3, NH4+, CH3+ D) CH3+, NF3, NH4+, XeF4
11) What is the bond angle in the following molecular model of NO2-? A) less than 109.5 B) 109.5 C) less than 120° but greater than 109.5 D) 120° 12) 12) What are the bond angles in the folowing molecular model of SO32-? ao A) less than 109.5 B) 109.5° C) less than 120° but greater than 109.5 D) 120 13) 13) How many of the following molecules are polar? B) 0 D) 3 E) 1 A) 2 14 14) Determine the electron geometry (eg, molecular geometry (mg. and polarity of Pa A) eg-trigonal pyramidal mg-trigonal pyramidal polar B) eg-tetrahedral mg-bent, polar C) eg-tetrahedral, mg-trigonal pyramidal polar D) eg-linear, mg-linear, nompolar E) eg-trigonal planar, mg-trigonal planar, nonpolar 15) Which orbital hybridization is associated with a tetrahedral charge cloud arrangement? 15)__ B) sp3 D) None of these C) sp 16) Which of the following is not true? 16 A) An sp3 hybrid orbital may hold a lone pair of electrons B) An sp3 hybrid orbital may form a pi bond by overlap with an orbital on another atom. C) An sp3 hybrid orbital may form a sigma bond by overlap with an orbital on another atom. D) The sp3 hybrid orbitals are degenerate.
chlo 17) What orbital hybridization is expected for the central atom in a molecule with a trigonal 17)_ planar geometry? A) sp2 C) sp B) sp3 D) None of these 18) Which atomic orbitals are involved in bonding and which as lone pair orbitals for 18) N2H2? H N N-H A) boading: s on H, sp2 and p on N B) bonding: s on H, sp2 on N C) bonding: sp2 on both H and N D) bonding: s on H, p on N lone pair: sp2 on N lone pair: p on N lone pair: p on N lone pair: sp2 on N 19) Place the following in order of increasing dipole moment 20) Give the hybridization for the O in OF2 21) Give the hybridization for the Br in BrCl3 22) Give the hybridization for the C in HCCH A)sp 20) E) sp3 Ð)sp3d 21) D) sp E)sp3 A)sp3 B) sp3d2 C) sp 22) D) sp3d E)sp2
23) Identify the set of hybrid orbitals shown below. A) sp C) sp3 B) sp2 D) None of these 24) Which molecule has a central atom that uses the set of hybrid orbitals shown below to form bonds 24) with the non-central atoms? A) NO2 B) H20 C) co2