1) Draw a complete arrow pushing mechanism for the pivaloylation experiment. Use 'R-OH' as the alcohol in your mechanism.
2) Draw a "to-scale" replica of thge TLC plate that you will be preparing for the competition experiment. Which would you expect to be found higher up on the silica plate during TLC analysis -- the alcohol strating material or the ester product? Explain your answer.
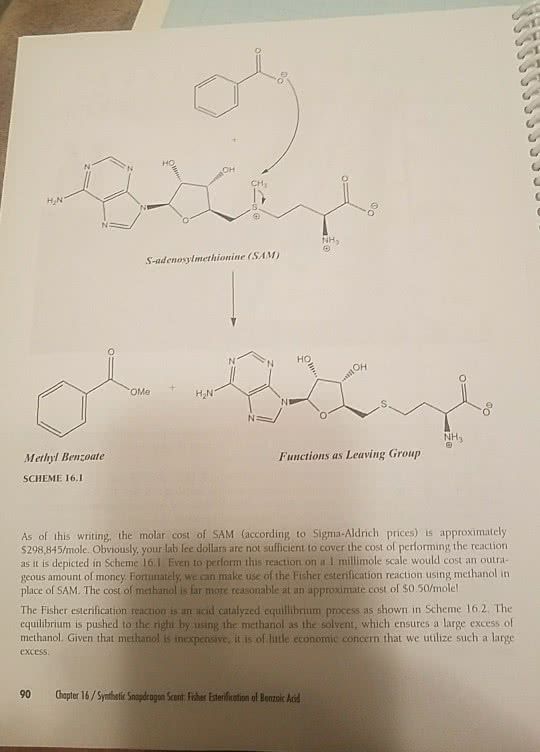
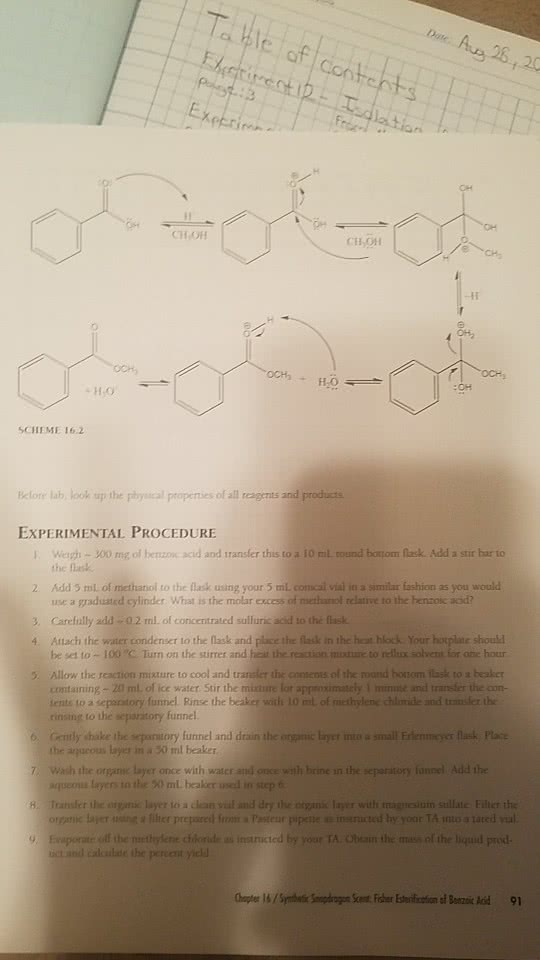
THEORY Esters can be made by a variety of reactions. The acetate synthesis performed in this lab m illustrates the most fundamental method of preparing esters, the Fischer esterification corresponding to the general equation: H SO R2-OH R1 OH R1 OR When a carboxylic acid and an alcohol are reacted together in this manner, a strong mineral acid (for example, HSO is usually added to catalyze the reaction. R and R can be any organic group (alkyl alkenyl, alkynyl, ryl) and they do not have to be the same. n-Butyl acetate is added to a variety of commercial products to create fruit flavours, especially apple, pear, and banana. This ester can also be used as a solvent for laque plastics, leather finishing, rubber and camphor The "Competition Experiment" section of this lab provides an example of another method of making esters the reaction of alcohols with carboxylic acid chlorides. UNDERSTANDING THE ExPERIMENT the two week period since your last lab, the este has been hopefully will have gone to completion. To separate the desired product from unreacted starting materials and unwanted side products, a distillation is performed. The distill product contaminated with trace amounts of acid and, thus, a neutralization step and drying M to remove traces of water are required to with obtain reasonably pure product. In the competition experiment, two different alcohols will compete for pivaloyl chloride in the esterification reaction. Which alcohol will win? analysis of the reaction will reveal the answer.