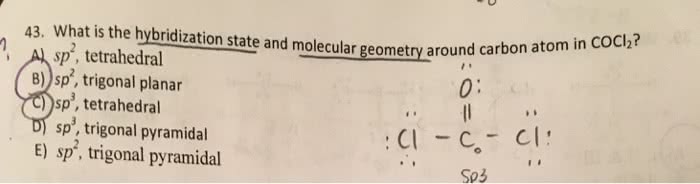CHMB41H3 Final: practice exam
Document Summary
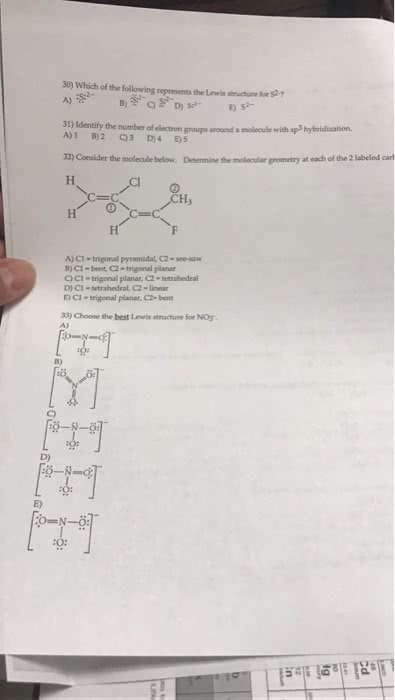
Part i (10 marks) - multiple choice (1 mark each) place your answers in the boxes on the answer sheet: the structure of doxorubicin, a drug widely used in chemotherapy and treatment of cancer, is shown below. Answer questions 7 to 9 using one of the following 6 terms. (a) constitutional isomers (d) enantiomers (b) identical molecules (e) not related (different) (c) cis/trans or e/z isomers. Br: which is the correct iupac name for the structure shown on the right, 5-sec-butyl-4-cyclopropyl-7-methylnonane, 5-sec-butyl-6-cyclopropyl-3-methylnonane, 4-sec-butyl-5-cyclopropyl-2-ethyloctane, 5-sec-butyl-4-cyclopropyl-7-ethyloctane, 5-sec-butyl-7-ethyl-3-methylcyclopropane. Part ii: short answers (i) on the answer sheet, complete the partial structures for the two chair conformations of cis-1- bromo-4-sec-butylcyclohexane. (ii) on the answer sheet, complete the partial structures for the two chair conformations of trans- 1- bromo-4-sec-butylcyclohexane. (iii) designate which conformation (a or b) is more stable in both part (i) and (ii). (circle one for each on the answer sheet).