BIOC 212 Chapter Notes - Chapter 6-7: Demand Curve, Perfect Competition, Commodity Market
Get access


Related Documents
Related Questions
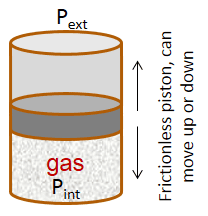
| pV work - Exercise 1 Consider a container with a frictionless piston that contains a given amount of an ideal gas. If the external pressure is kept constant, the piston will move up or down in response to a change in the internal pressure. The piston will move up if Pint > Pext and vice versa. The piston will stop moving when Pint = Pext (the system is equilibrated).
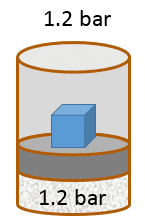
The following problem will slowly guide you through a work calculation. Some questions may appear trivial, so trust your instincts and donât assume there are hidden tricks. The goal of such a guided exercise is to help you picture the situation so you understand how all relevant variables change or stay the same. This is the first in a sequence of related problems. Remember to use three significant figures for all numerical answers. The margin of error for each numerical answer is 5%. To avoid rounding errors use the unrounded intermediate values in your final calculations. Letâs assume the initial volume of the gas is 3.90 L , the initial temperature of the gas is 26.0 °C , and the system is in equilibrium with an external pressure of 1.2 bar (given by the sum of a 1 bar atmospheric pressure and a 0.2 bar pressure due to a brick that rests on top of the piston).
| Part A How many moles of gas are there in the container?
SubmitMy AnswersGive Up You now heat the gas slowly until the temperature reaches 51.2 °C You do it slowly so the piston can move if necessary until a new equilibrium is reached. Part B What happens with the piston while you heat the gas? What happens with the piston while you heat the gas?
When the final temperature is reached, you stop heating the gas. Part C What is the final pressure of the gas?
SubmitMy AnswersGive Up Part D What is the final volume of the gas?
SubmitMy AnswersGive Up Part E What happens with the piston after you finish heating the gas? Assume you do not need to worry about the gas cooling down again because the outside of the container is at a lower temperature. That is, you manage to keep the gas at a constant temperature that equals 51.2 °C What happens with the piston after you finish heating the gas? Assume you do not need to worry about the gas cooling down again because the outside of the container is at a lower temperature. That is, you manage to keep the gas at a constant temperature that equals 51.2
SubmitMy AnswersGive Up So far this was all at the level of your general chemistry class. Letâs now calculate the work involved in the process. Recall the sign conventions we use in physical chemistry, and always think from the point of view of the system (the gas). Part F What is the sign of w? What is the sign of w?
SubmitMy AnswersGive Up Part G What is the value of w? Be careful with units. How do you convert bar*L to J?
SubmitMy AnswersGive Up |
Material Science
Question 1
Muscle fibers slide smoothly across each other in both directions.
True
False
Question 2
Cell membranes are composed of single layers of fat molecules.
True
False
Question 3
In mammals, the percentage composition of fats and minerals is roughly the same.
True
False
Question 4
Both RNA and DNA have a double helix structure.
True
False
Question 5
The primary constituents of muscles are
| a) | the proteins kerotin and actin | |
| b) | the proteins lycine and actin | |
| c) | the proteins actin and liposine | |
| d) | the proteins glycine and lycine | |
| e) | the proteins myocin and actin |
Question 6
Hydroxyapatite is a component of endoskeletons.
True
False
Question 7
There are thousands of types of amino acids.
True
False
Question 8
Amorphous silicon is a component of some biomaterials.
True
False
Question 9
The biosynthesis of both collagen and polysaccharides starts in the cell nucleus.
True
False
Question 10
After going through alkyline hydrolysis, the remaining solids are primarily hydroxyapatite.
True
False
Question 11
Calcium carbonate is a mineral found in some biomaterials.
True
False
Question 12
Proteins, chitin, and cellulose are build up from proteins
True
False
Question 13
There are thousands of types of proteins.
True
False
Question 14
Chitin is the most abundant natural polymer.
True
False
Question 15
Keratin is formed from a structure of living cells.
True
False
Question 16
Some biopolymers are hard.
True
False
Question 17
Polymeric biomaterials are comprised of collagen and elastin.
True
False
Question 18
Cell membranes are made of double layers of proteins.
True
False
Question 19
Which is NOT an example of a hard biopolymer?
| a)Eggs | ||
| b)Insect Shells | ||
| c)Hooves | ||
| d)Nails | ||
| e)Cellulose |
Question 20
Polysaccharides are built up hierarchically from amino acids.
True
False
Question 21
Unit cells have faces that are
| a) | rhombus | |
| b) | square | |
| c) | rectangular | |
| d) | parallelegrams | |
| e) | circular |
Question 22
Unit cells are
| a)the building blocks of crystalline material | ||
| b)the building blocks of DNA | ||
| c)the building blocks of polymers | ||
| d)the building blocks of amorphous materials | ||
| e)the building blocks of biomaterials |
Question 23
Which radioactive semi-metal is the only element with a Simple-Cubic (SC) structure?
| a)Polonium | ||
| b)Uranium | ||
| c)Radium | ||
| d)Plutonium | ||
| e)Dilthium |
Question 24
The common crystal structures in metal are
| a)FCC, BCC, HCP | ||
| b)FCC, BCC, HCP, and SC | ||
| c)HCC, BCC, FCP | ||
| d)HCC, BCC, FCP and SC | ||
| e)FCC, SEC, BCC |
Question 25
Diffraction results in
| a)constructive interference | ||
| b)delamination of the planes | ||
| c)reverse polarization | ||
| d)altered planar spacing | ||
| e)destructive interference |
Question 26
Unit cell indexing schemes provide
| a)location information for grain boundaries | ||
| b)descriptions of the atoms, planes, and axis directions | ||
| c)diffraction indexes for the crystal material | ||
| d)the location of the unit cell in the larger crystalline matrix | ||
| e)the packing arrangement |
Question 27
Hexagonal crystal structures cannot be close-packed.
True
False
Question 28
To determine crystal structure and spacing, scientists use
| a)magnetic resonance imaging | ||
| b)X-ray diffraction | ||
| c)optical microscopes | ||
| d)scanning electron microscopes | ||
| e)atomic force microscopes |
Question 29
A scanning probe (aka atomic force) microscope can be used to image and manipulate individual atoms.
True
False
Question 30
Crystal planar density is taken as
| a)the number of atoms per unit area for a plane | ||
| b)the planar stacking order | ||
| c)the number of atoms that are closely packed | ||
| d)the number of equivalent atoms per unit cell | ||
| e)the number of atoms per unit length for an axis |
Question 31
The atomic packing factor represents
| a)the mass ratio of the unit cell | ||
| b)the number of nearest-neighbor atoms | ||
| c)the fraction of solid sphere volume in the unit cell | ||
| d)the unit cell edge length (a) divided by the atomic radius (R) | ||
| e)the density of the material |
Question 32
When an elemental solid can have more than one crystal structure, it is said to have
| a)allotropy | ||
| b)polymorphism | ||
| c)polytropy | ||
| d)elemorphism | ||
| e)anthropomorphis |
Question 33
The crystallographic linear density
| a)is determined by the atomic packing factor | ||
| b)is six for HCP unit cells | ||
| c)is five for BCP unit cells | ||
| d)is determined by the coordination number | ||
| e)is how many atoms lie along a unit length in that direction |
Question 34
Bragg's law specifies
| a)the condition for when electrons will be refracted upon striking a crystal | ||
| b)the condition for when light will be polarized upon striking a crystal | ||
| c)the condition for when x-rays will be absorbed upon striking a crystal | ||
| d)the condition for when x-rays will be refracted upon striking a crystal | ||
| e)the condition for when light will be refracted upon striking a crystal |
Question 35
Two features of a crystal unit cell structure are
| a)atomic number and elemental packing factor | ||
| b)the number of interstitial and host atoms | ||
| c)coordination number and atomic packing factor | ||
| d)the number of faces and the number of atoms | ||
| e)the number of substitutional and interstitial atoms |
Question 36
Imperfections in semiconductors are used to create transistors and other electronic devices.
True
False
Question 37
The majority of crystalline solids are single crystals
True
False
Question 38
The grain structure of a polycrystalline material can be observed in the material without significant preparation.
True
False
Question 39
There are three types of electron microscopes, Transmission (TEM), Scanning (SEM), and Tunneling (TUEM).
True
False
Question 40
Defects are important for solid catalysts
True
False
Question 41
The mean intercept length is a measure of grain diameter.
True
False
Question 42
Imperfections in materials do not generally change their properties.
True
False
Question 43
The mean intercept length is computed from the average length of a grain boundary edge.
True
False
Question 44
Catalysts chemically combine with the reactant molecules in order to speed up the reaction.
True
False
Question 45
For a particular material, the higher the temperature the greater the number of vacancies.
True
False
Question 46
A twin boundary is a linear defect where the screw angles on each side of the boundary are equal.
True
False
Question 47
The regular pattern of crystal order is disrupted at a grain boundary.
True
False
Question 48
Solid solutions can only be formed by liquid material cooling to a solid.
True
False
Question 49
Alloys do not usually have significantly difference properties of the constituent metals.
True
False
Question 50
When an atom is missing from a crystal structure position, it is referred to as a self-interstitial.
True
False