BCH 261 Chapter 3: Biochemistry Chapter 3 (Lecture 4 and 5) Energetics
Document Summary
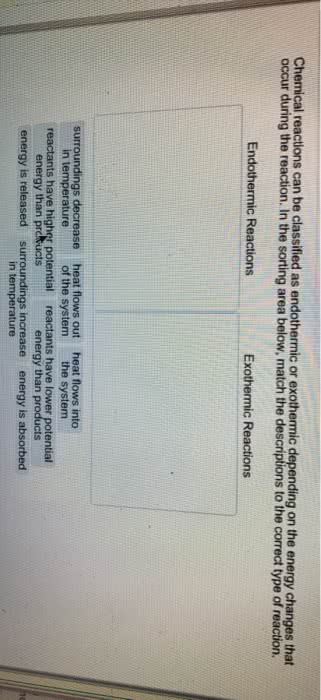
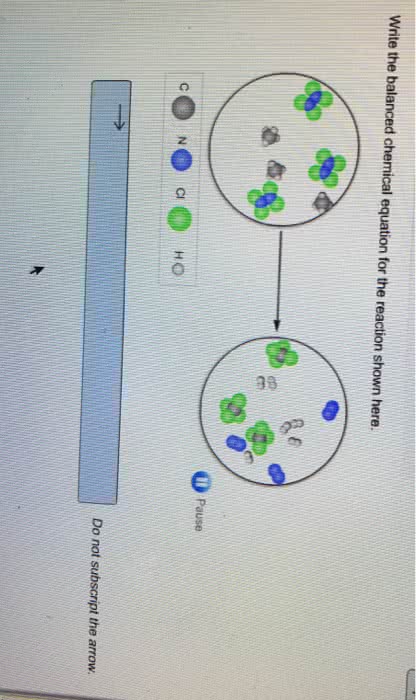
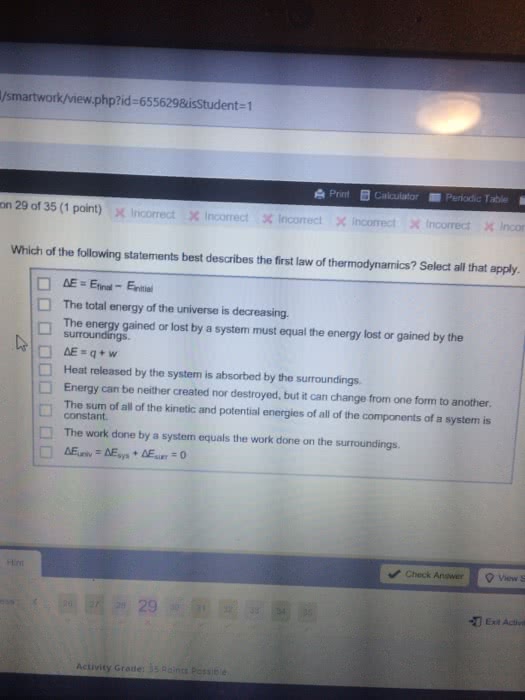
Free energy change ( g) can determine whether a process/reaction will require or release energy and how much energy will the process make that is able to do useful work. System- any part of the universe that we choose for study. Surroundings- anything that is not defines as part of the system. States that energy is conserved in a closed system. Energy cannot be created or destroyed, it can only be transformed from one type of energy to another. Enthalpy (h)- the amount of energy released or absorbed by the system in the form of heat (total heat content of a system) H = the reaction releases energy in the form of heat = exothermic reaction = spontaneous. + h = the reaction absorbs energy in the form of heat = endothermic reaction = non-spontaneous. State function- a property whose value does not depend on the path taken to reach that specific value.