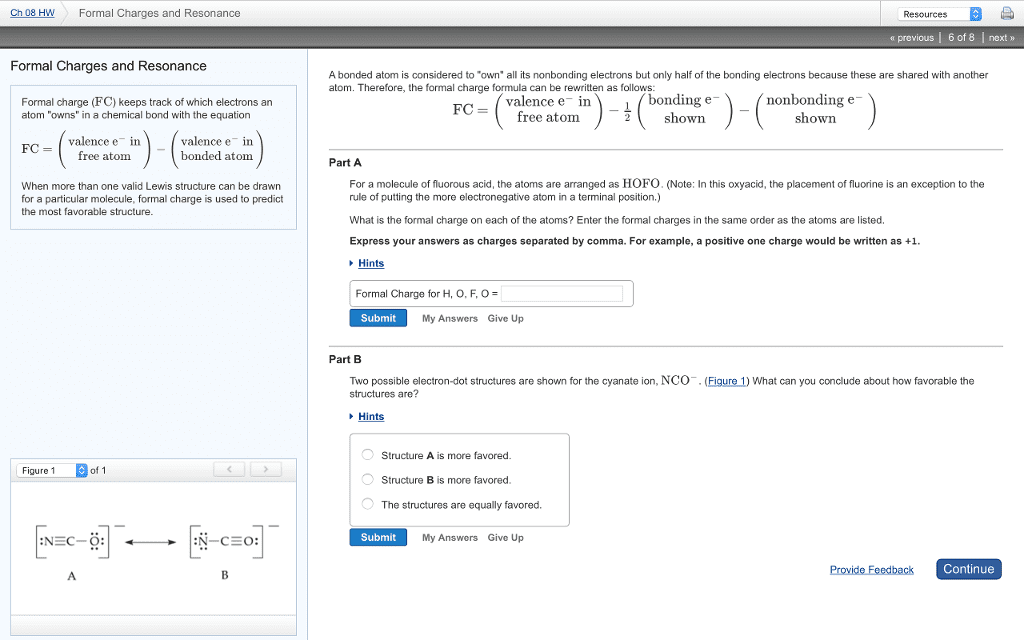
CHEM 110 Chapter Notes - Chapter 9: Ionic Compound, Potassium Bromide, Joule

Chapter 9 Chemical Bonding
- Lewis theory
o Valence electrons are represented as dots and we can draw Lewis electron-dot
structures
▪ Lewis theory can be used to preduct whether a particular set of atoms will
form a stable molecule and what that molecule might look like
- Types of Chemical Bonds
o Atoms are composed of particles with Positive charges and negative charges
o Ionic, Covalent and Metallic Bonding
▪ When a metal bonds with a non-metal it transfers one or more electrons to
the non-metal (Ionic bond)
▪ When a nonmental bonds with another non-metal, the two atoms share
electrons (Colvalent bond)
▪ Negatively charged particles interacts most strongly with both of the
positively charged ones
• Negatively charged particles hold two positively charged ones
together
• Hold the bonding atoms together by attracting positive charges
▪ Third type of bonding occurs in metals.
• Metals tend to lose electrons easily, all of the atoms in a metal
lattice pool their valence electrons
• Positive metals atoms are attracted to seao f electrons, holding
metal together
- Representing valence electrons with Dots
o Valence electrons are those electrons in the
outmost principal energy level
o Valence electrons are held most loosely,
and are the most important in bonding
o In a lewis structure we represent the valence
electrons of main group elements as dots
surrounding the symbol for the element
▪ Each dot represents a valence electron
▪ Dots are placed around the elements symbol with two dots per side
▪ Provides a simple way to visualize the number of valence electrons in a
main group atom
• With eight valence electrons, they have a full outer level
o Octet
• Helium: two electrons represents a stable electron because N =1
o A chemical bond is the sharing or transfer of electrons to attain a stable electron
configurations for the bonding atoms
▪ If the electrons are transferred, the bond is ionic
▪ If electrons are shared, the bond is covalent
▪ When applying Lewis theory, we do not calculate energies associated with
attractions and repulsions between electrons and neighbouring atoms
find more resources at oneclass.com
find more resources at oneclass.com

• Lewis Theory uses simple octet rule, that predicts what we see in
nature for a large number of compounds
- Ionic Bonding and Lattice Energies
o Lewis theory can also be applied to ionic bonding
o To represent ionic bonding, we move electron dots from Lewis structure of the
metal to the Lewis Structure of the non-metal
▪ Creating a crystalline Lattice composed of alternating cations and Anions
▪ The transfer of the electrons gives chlorine an octet and leaves potassium
without any valence electrons but with an octet in previous principal
energy level
• The potassium is positively charged because it loses an electron
o While the chlorine, is negatively charged because it gains
an electron
▪ Creating a crystalline with Na & S
• Sodium must lose its one valence in
order to have an Octet while sulfur
must gain two electrons to get an octet
o Two sodium atoms each lose their valence electron while
sulfur gains two electr ons and gets an octet
▪ Na2S
o Lattice Energy
▪ When sodium Chloride forms from elemental sodium and chloride, 411 kJ
of heat is evolved in the following reaction
• Na(s) + ½ Cl2 NaCl (s) Δf H = -411kJ Mol
o The transfer of an electron from sodium to chlorine actually
absorbs energy
o First ionization of sodium is 496kJ mol-1 and affinity of Cl
is 349kJ mol-1
▪ Why is the reaction exothermic
• Lattice Energy
o Energy associated with forming a
lattice of alternating cations and
anions from gaseous ions
o Since sodium is positively charged
and chlorine is negative, the potential
energy decreases
o Energy is emitted as heat when the
lattice forms
o Easiest way to calculate lattice
energy is with Born-Haber cycle
o Born-Haber Cycle
▪ Series of steps that represents the formation of an ionic compound from its
elements
find more resources at oneclass.com
find more resources at oneclass.com

▪ The steps are chosen so change in enthalpy of each step is known except
for the last one (Lattice energy)
▪ Using Hess’s law we can determine the enthalpy change for unkown last
step
• Example: NaCl
o Na(s) + ½ Cl2 NaCl (s) Δf H = -411kJ Mol
• First step is the formation of gaseous sodium from solid sodium
o Na (s) Na (g) = 108kJ mol-1
• Second Step is formation of chlorine atom from chlorine molecule
o ½ Cl2 Cl (g) = 122kJ mol-1
• Third Step is the ionization of gaseous sodium
o Na (g) Na+ (g) + e- = 496kJ mol-1
• Fourth Step is addition of electron to gaseous chlorine
o Cl (g) + e- Cl- (g) = -349kJ Mol-1
• Fifth step is the formation of Crystalline solid from Gaseous
o Na+ (g) + Cl- NaCl (s) = ?
• Δf H = Δr H1 + Δr H2 + ΔrH3 + ΔrH4 + ΔrH5
o ΔrH5 is Lattice energy
• ΔrH5 = -411kJ mol – (108kJ + 122kJ) + 496kJ – 349 kJ
o -788kJ Mol-1
▪ The magnitude of the Lattice energy of the chlorides decreases
accordingly, making the formation of chlorides less exothermic
• As the radii increase when we move down the column, the ions
cannot get as close to each other and therefore do not release as
much energy
• Why is the magnitude of CaO greater than that of NaF
o Na has a radius of 95pm and F has a radius of 136
o Ca2+ has a radius of 99pm and O2 of 140pm
o Summary
▪ Lattice eneriges become less exothermic with increasing ionic radius
▪ Lattice energies become more exothermic with increasing magnitude
o Example: Arrange these compounds in order of increasing magnitudes of lattice
energy
▪ CaO, KBr, KCl, SrO
• KBr and KCl have lattice energies of smaller magnitude than CaO
and SrO because of their lower ionic charges
o (1+, 1- compared to 2+, 2-)
• KBr is expected to have a lower magnitude due to the radius of the
bromide ion relative to chloride
• SrO has a lower magnitude due to the radius of the strontium to
calcium
- Covalent Bonding: Lewis Structures of Molecules
o Hydrogen does not obey octet rule because to fill its valence shell. It requires only
two electrons
o Hydrogen is stable with two electrons
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
If the electrons are transferred, the bond is ionic. Covalent bonding: lewis structures of molecules calcium: hydrogen does not obey octet rule because to fill its valence shell. It requires only two electrons: hydrogen is stable with two electrons. If any nonhydrogen atoms lack an octet, form double or triple bonds to give them octets. Bond polarity, dipole moment and percent ionic character: degree of polarity in a chemical bond depends on electronegativity difference between two bonding elements, greater the difference, the more polar the bond. It was previously calcaulated that separating a proton and electron by 130pm results in a dipole of 6. 2d: 3. 5d/6. 2d * 100% = 56% In lewis theory, electrons are localized on one atom or between them: the delocalization of electrons lowers their energy, stabilizing them, depicts two or more structures with the electrons in different palces to accurately reflect delocalization of electrons.