CHEM392A Chapter Notes - Chapter 5: Potential Energy, Thermal Energy, Intermolecular Force
Document Summary
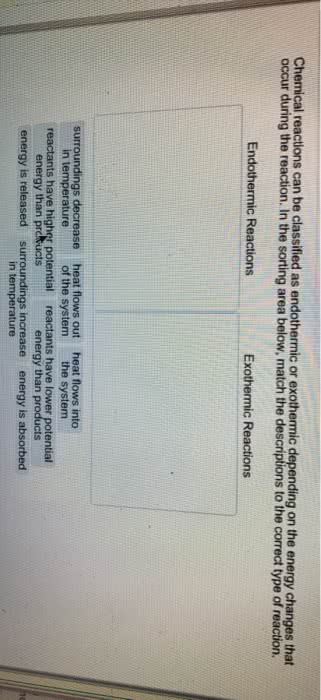
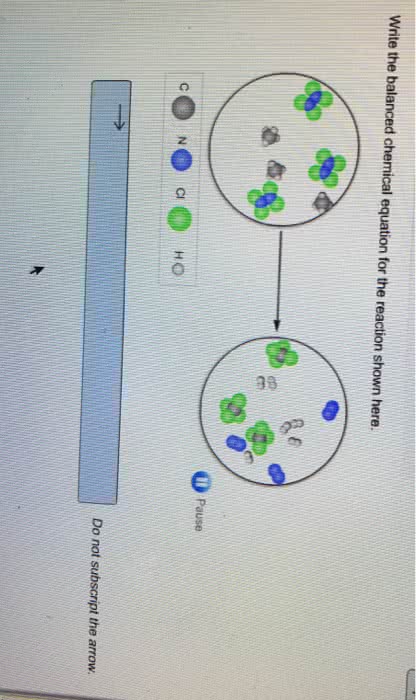
Thermochemistry: the study of the energy changes that accompany physical or chemical changes in matter. Thermal energy: energy available from a substance as a result of the motion of its molecules. Chemical system: a set of reactants and products under study, usually represented by a chemical equation. Surroundings: all matter around the system that is capable of absorbing or releasing thermal energy. Heat: the amount of energy transferred between substances , q. Exothermic: releasing thermal energy as heat flows out of the system. Endothermic: absorbing thermal energy as heat flows into the system temperature: average kinetic energy of the particles in a sample of matter. Open system: one in which both matter and energy can move in or out (ex. Isolated system: an ideal system in which neither matter nor energy can move in nor out (cannot completely isolate!) Closed system: one in which energy can more in or out, but not matter.