CHMB42H3 Chapter 17: CHMB42 Chapter 17
Document Summary
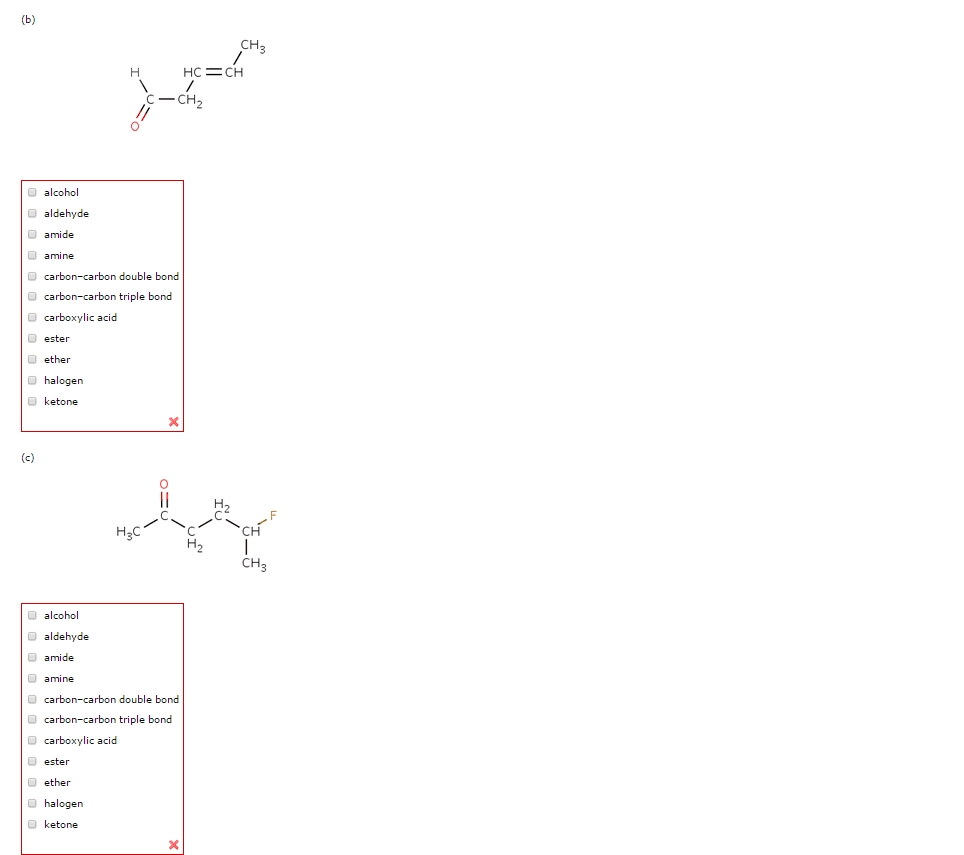
The systematic name of an aldehyde is obtained by replacing the final e on the name of the parent hydrocarbon with al (ethanal, 2-bromopropanal or methanal). Terminal e is removed only to avoid two successive vowels (hexanedial, e isn"t removed). If a compound has two functional groups, the one with the lower priority is indicated by a prefix and the one with the higher priority by a suffix. The systematic name of a ketone is obtained by replacing the final e on the end of the name of the parent hydrocarbon with one. The chain is numbered in the direction that gives the carbonyl carbon the smallest number (3-hexanone, 6-methyl-2-heptanone or acetone). If the ketone has a second functional group of higher naming priority, the ketone oxygen is indicated by the prefix oxo. Increasing priority: alkyl halide < ether < alkane < alkyne < alkene < amine