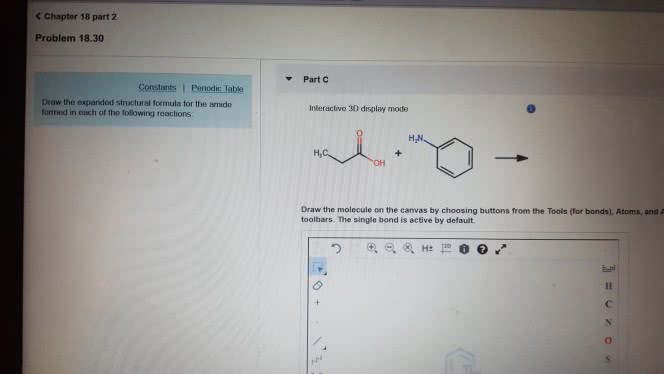
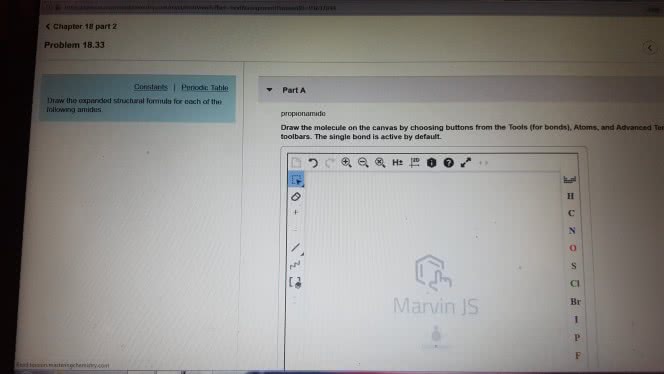
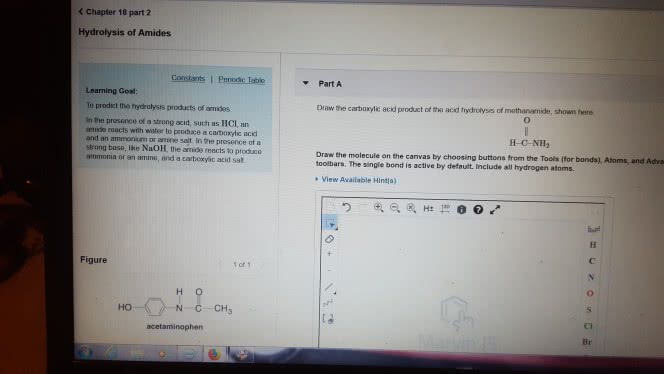
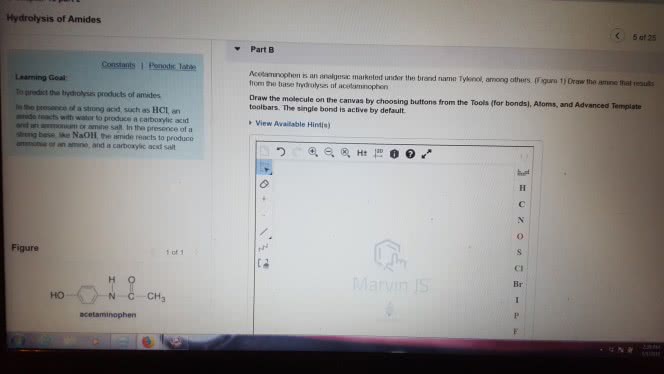
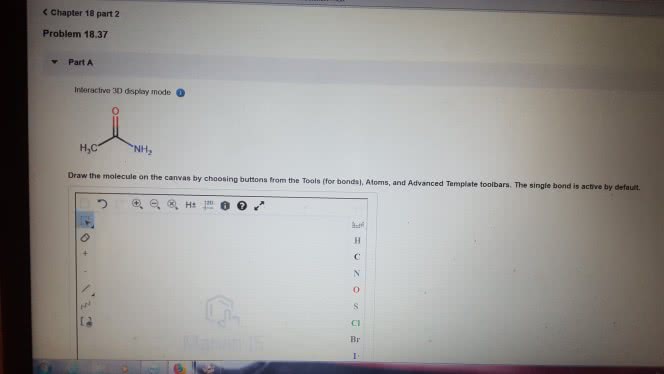
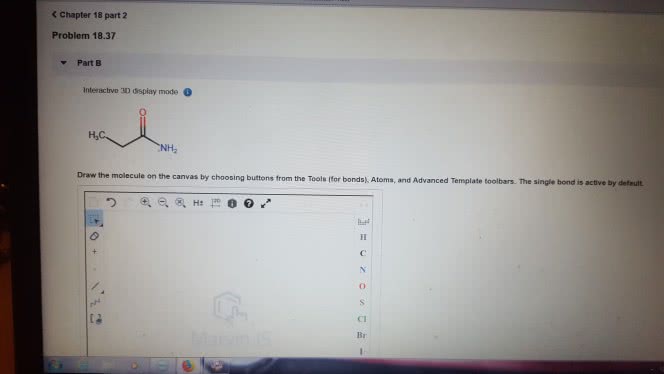
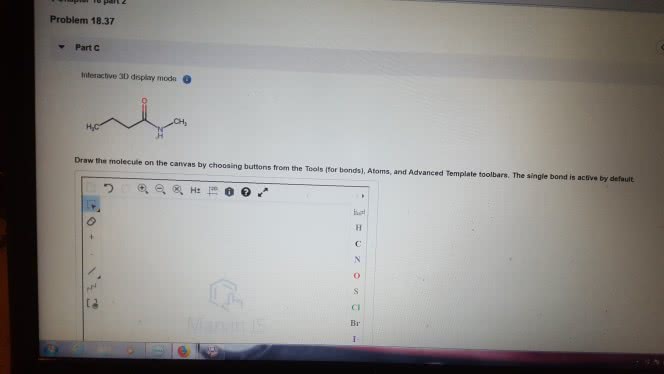
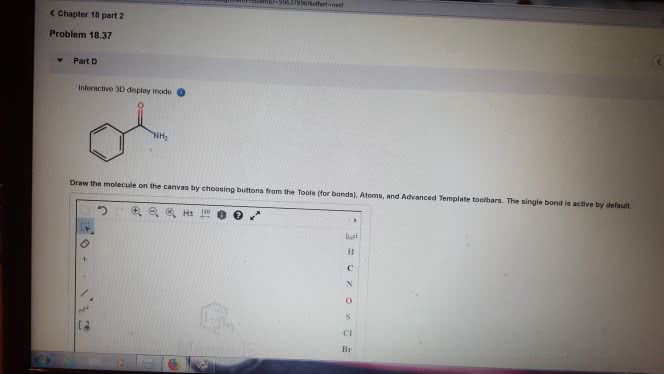
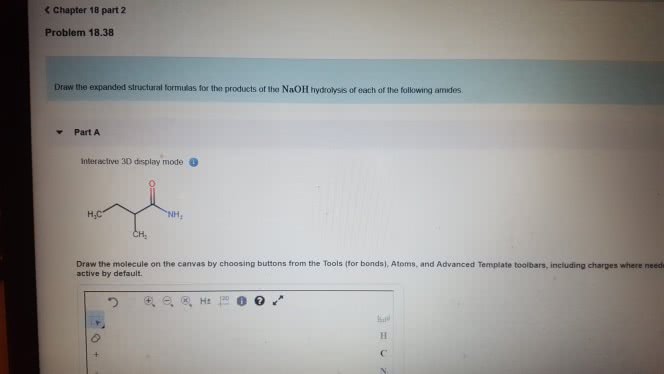
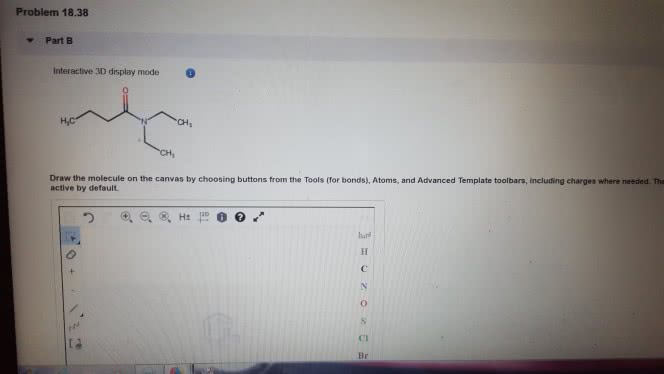
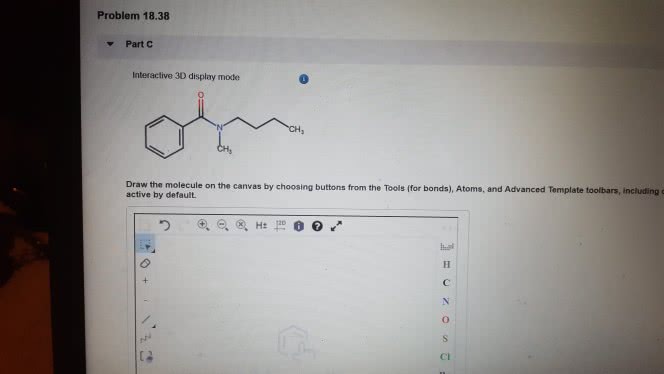
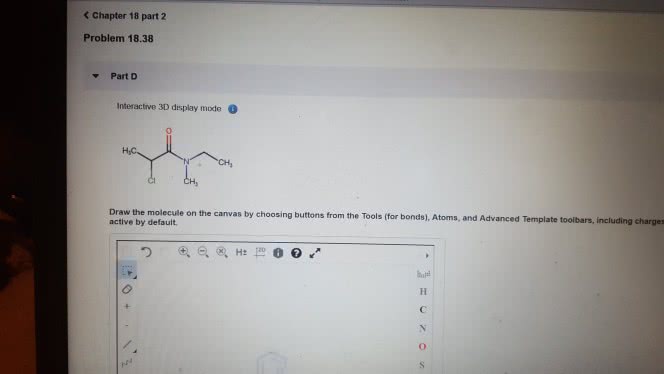
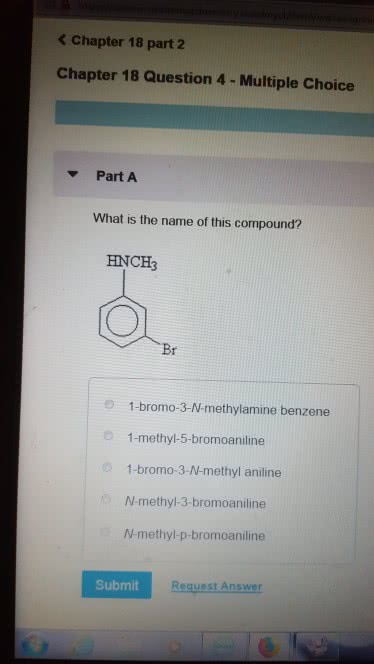
1)The reaction of benzamide, C6H5CONH2, with LiAlH4 yields which of the following compounds?
A.benzoic acid
B.benzonitrile
C.benzylamine
D.ammonium benzoate
E. none of the above
2) Electron-releasing groups bonded to an amine nitrogen increases it basicity. T or F?
3) Ammonium salts are water soluble. T or F?
4)A compound which is insoluble in water but which dissolves in dilute hydrochloric acid is
A. probably an amide
B.possibly a carboxylic acid
C.almost certainly an amine
D.probably neither acidic nor basic
E.probably an ester
5)Primary and secondary amines react with nitrous acids to form stable diazonium salts. T or F?
6). Dimethylethylamine is classified as a
A.primary amine
B.secondary amine
C.tertiary amine
D.quaternary amine
7)Heating a(n) _______ results in a Cope elimination.
A.amine oxide
B.imine
C.enamine
D.oxime
E.quaternary ammonium salt
8) Aniline is less basic than methylamine because:
A. resonance in aniline increases the electron density on nitrogen: this effect is not possible for methylamine.
B.resonance in aniline stabilizes the nonprotonated base but does not stabilize the protonated base: in methylamine resonance is of no importance for the protonated or unprotonated forms.
C.the acid-base reaction of aniline with a proton is kinetically rather than thermodynamically controlled.
D.the ortho hydrogens of aniline interfere with protonation of its amino group.
E.The question is incorrect, aniline is actually more basic than methlamine.
9) Amines can best be separated from nitro compounds by an extraction procedure employing diethyl ether and:
A.water
B.aqueous HCl
C.aqueous NaOH
D.concentrated H2SO4
10) An amino group is a powerful ortho-para director. T or F?
11) What is the hybridization of the nitrogen atom in CH3NH2?
A. sp
B.sp2
C.sp3
D.s-sp3
E.a primary amine
12) Diazonium salts readily couple with phenols and aromatic amines to form azo compounds. T or F?
13) Aniline is a stronger base than ammonia. T or F?
14) What type of amine is tert-butyl amine?
A.primary
B.secondary
C.tertiary
D.quaternary
E.none of these