01:160:307 Chapter Notes - Chapter 8: Regioselectivity, Bromine, Halogenation
Document Summary
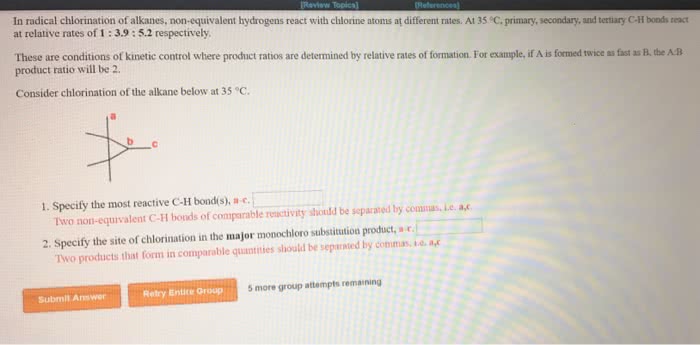
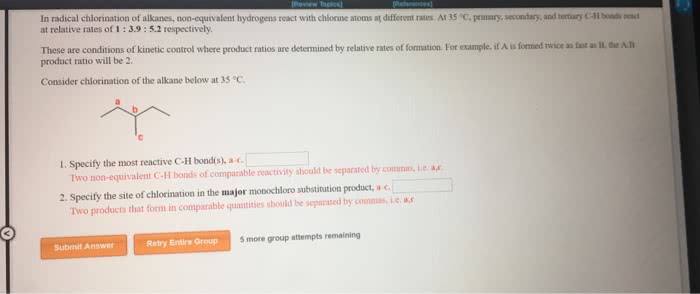
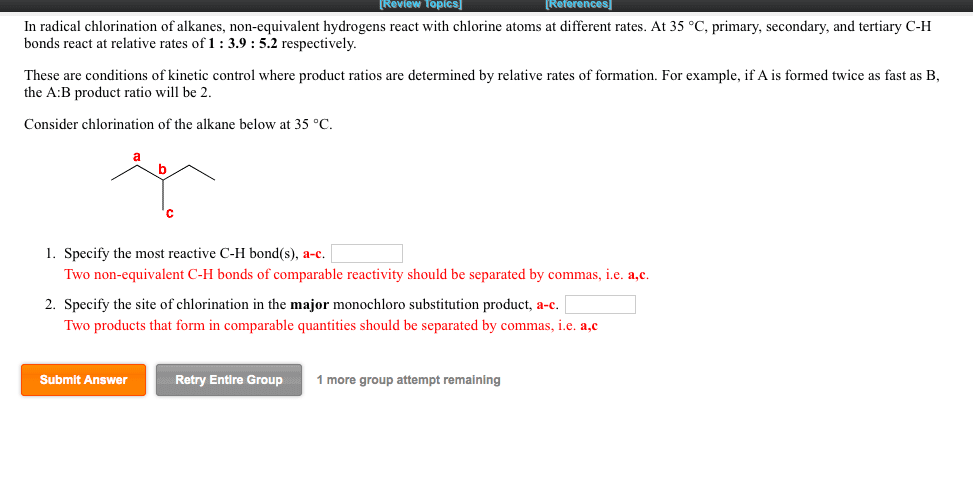
Regioselectivity: for both reactions, there were 2 types of h"s the halogen could have replaced: 6 primary. 3o h"s: ratio of 2o h"s * # of eq. 2o h"s: ratio of 1o h"s * # of eq. 3o h"s = 5 * 0 = 0: ratio of 2o h"s * # of eq. 2o h"s = 4 * 2 = 8: ratio of 1o h"s * # of eq. 3o h"s = 1600 * 0 = 0: ratio of 2o h"s * # of eq. 2o h"s = 80 * 2 = 160: ratio of 1o h"s * # of eq. 1:2:5, determine the relative yield of each monochlorination product formed: ignoring stereoisomers, draw the structures of all possible monobromination products that form when the following molecule reacts with bromine and light. Given that the reactions of 1 , 2 , and 3 c h bonds proceed with a selectivity of.