CHEM1006 Lecture Notes - Lecture 18: Sigma Bond, Octet Rule, Pi Bond
Document Summary
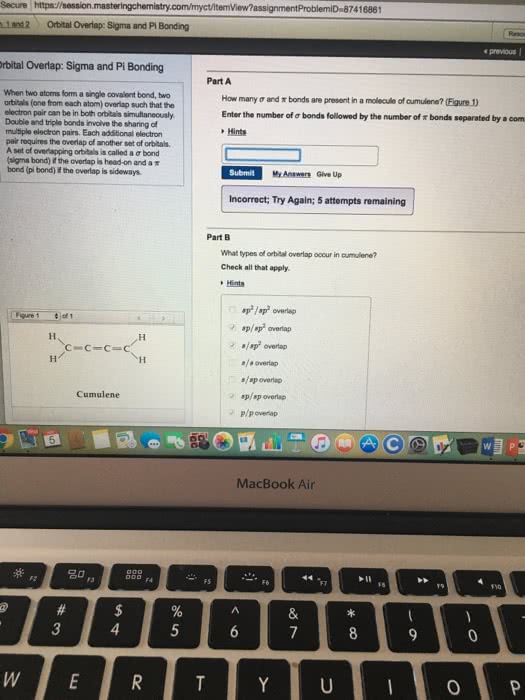
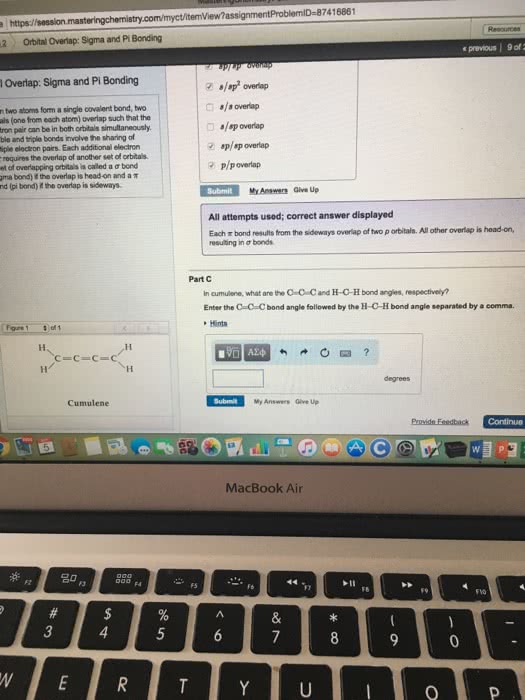
Covalent bonds are a class of chemical bonds where valence electrons are shared between two atoms, typically two nonmetals. The formation of a covalent bond allows the nonmetals to obey the octet rule and thus become more stable. If it shares one electron with a carbon atom, the fluorine will have a full octet. Covalently sharing two electrons is also known as a. Carbon will have to form four single bonds with four different fluorine atoms to fill its octet. Covalent bonding requires a specific orientation between atoms in order to achieve the overlap between bonding orbitals. Sigma bonds are the strongest type of covalent interaction and are formed via the overlap of atomic orbitals along the orbital axis. The overlapped orbitals allow the shared electrons to move freely between atoms. Pi bonds are a weaker type of covalent interactions and result from the overlap of two lobes of the interacting atomic orbitals above and below the orbital axis.