PCL201H1 Lecture 4: Lecture 4 Drug-Receptor Interaction
Document Summary
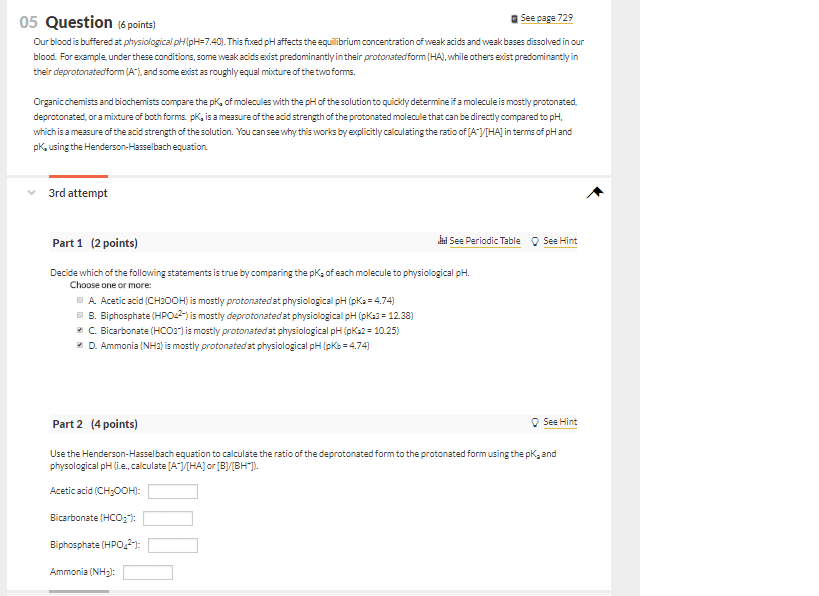
Ionization is particularly important, our physiological system is aqueous, water is abundant and can form dipoles and dissociate to interact with particular compounds. Ha h+ + a- acidic environment, forced to right weak acids will be protonated. Xh x + h+ deprotonated form more lipid soluble at low ph: rule of thumb: to roughly calculate percent ionization based on pka value of drug and ph of compartment. Ionization affects drug distribution: at physiological ph, morphine is ionized but fentanyl is not, so it can access brain more rapidly and is more potent, range of ph values in different compartments. Blood 7. 4 (we have an effective buffer system) Urine can change more substantially (5-8); can be changed by diet; vegetarians tend to have more basic urine. Stomach 2: compartment ph plays a role, protonated compound (charged), less lipid soluble. Ionization is important for absorption and distribution, reaching a particular target.