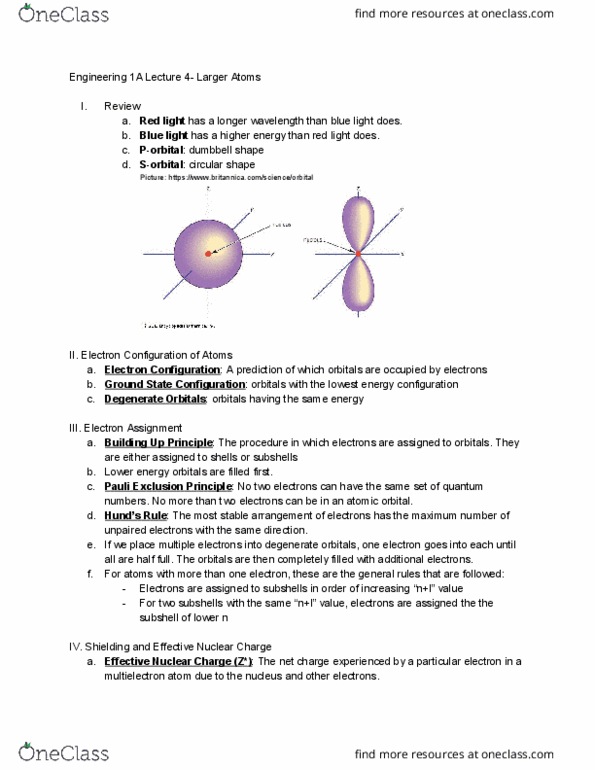
ENGR 1A Lecture Notes - Lecture 2: Metal, Atomic Number, Magnesium
ENGR 1A verified notes
2/31View all
Document Summary
Engineering 1a lecture 2- the periodic table and introduction to chemistry. 1b-8b are known as the transition elements: nonmetals : solids, gases and one liquid. Except for carbon, do not conduct: metalloids : have some properties that resemble metals and some that resemble electricity nonmetals. Group 1a: the alkali metals: all soft metallic solids, metallic luster when freshly cut b. Group 2a: the alkaline earth metals: magnesium and calcium are the 7th and 5th most abundant elements in the earth"s crust, react with water to form alkaline solutions. Group 3a: metalloids and metals: contains the first metalloids boron as well as aluminum, the most abundant element in. Earth"s crust: group 4a: non-metals, metalloids, and metals, first group containing non-metals, often form allotropes : elements that exist in different and distinct forms. Group 5a: contains nitrogen, which makes up about of the earth"s atmosphere, allotropes of phosphorus can react with air spontaneously. Group 7a: halogens: nonmetals that exist as diatomic molecules.