The chemicals that will be used in this experiment are: 2.0 x 10-4 M KSCN Potassium thiocyanate is the source of SCN- .
0.20 M Fe(NO3)3 Iron(III) nitrate is the source of Fe3+. Clean and dry 6 large test tubes, then label them #1 - #6. Use a 5 mL Pipetman to transfer 5.00 mL of 2.0 x 10-4 M KSCN solution into each test tube.
Each of the six test tubes should have 5.00 mL of potassium thiocyanate in it at this point. Test Tube #1 To the potassium thiocyanate solution in test tube #1, add 5.00 mL of the 0.20 M Fe(NO3)3 solution.
This solution will be used as the standard; SCN- is the limiting reactant, and the large excess of Fe3+ converts all of the SCN- into FeSCN2+. This sample will have the maximum concentration of FeSCN2+ complex and the most intense color.
Test Tube #2 Pipet 10.0 mL of the 0.20 M Fe(NO3)3 solution into a 25 mL graduated cylinder. Add distilled water to get 25.0 mL of solution. Mix thoroughly.
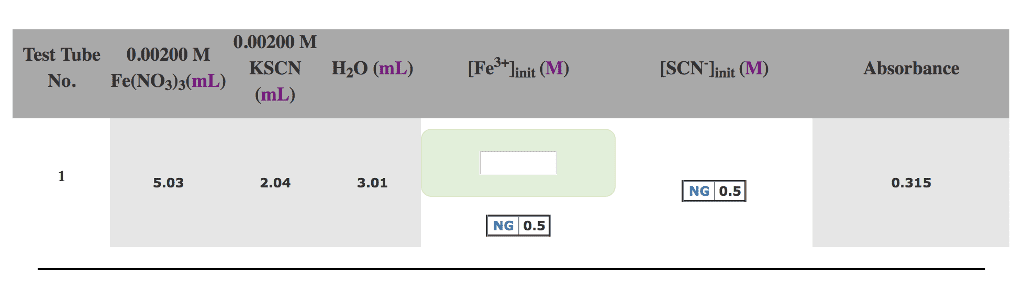
What is [Fe3+] in this 25.0 mL solution? Add 5.0 mL of the 25.0 mL Fe3+ solution to test tube #2. Remember that the tube already contains KSCN. What is [Fe3+]o for test tube #2? What is [SCN- ]o for test tube #2? Test Tube #3 Discard all but 10.0 mL of the Fe3+ solution in the graduated cylinder. To this 10.0 mL, add water to get 25.0 mL of solution.
Mix thoroughly. What is [Fe3+] in this 25.0 mL solution? Add 5.0 mL of the 25.0 mL Fe3+ solution to test tube #3. Remember that the tube already contains KSCN. What is [Fe3+]o for test tube #3? What is [SCN- ]o for test tube #3? Test Tube #4 Again, discard all but 10.0 mL of the Fe3+ solution in the graduated cylinder.
To this 10.0 mL, add water to get 25.0 mL of solution. Mix thoroughly. What is [Fe3+] in this 25.0 mL solution? Add 5.0 mL of the 25.0 mL Fe3+ solution to test tube #4. Remember that the tube already contains KSCN. What is [Fe3+]o for test tube #4? What is [SCN- ]o for test tube #4?
The chemicals that will be used in this experiment are: 2.0 x 10-4 M KSCN Potassium thiocyanate is the source of SCN- .
0.20 M Fe(NO3)3 Iron(III) nitrate is the source of Fe3+. Clean and dry 6 large test tubes, then label them #1 - #6. Use a 5 mL Pipetman to transfer 5.00 mL of 2.0 x 10-4 M KSCN solution into each test tube.
Each of the six test tubes should have 5.00 mL of potassium thiocyanate in it at this point. Test Tube #1 To the potassium thiocyanate solution in test tube #1, add 5.00 mL of the 0.20 M Fe(NO3)3 solution.
This solution will be used as the standard; SCN- is the limiting reactant, and the large excess of Fe3+ converts all of the SCN- into FeSCN2+. This sample will have the maximum concentration of FeSCN2+ complex and the most intense color.
Test Tube #2 Pipet 10.0 mL of the 0.20 M Fe(NO3)3 solution into a 25 mL graduated cylinder. Add distilled water to get 25.0 mL of solution. Mix thoroughly.
What is [Fe3+] in this 25.0 mL solution? Add 5.0 mL of the 25.0 mL Fe3+ solution to test tube #2. Remember that the tube already contains KSCN. What is [Fe3+]o for test tube #2? What is [SCN- ]o for test tube #2? Test Tube #3 Discard all but 10.0 mL of the Fe3+ solution in the graduated cylinder. To this 10.0 mL, add water to get 25.0 mL of solution.
Mix thoroughly. What is [Fe3+] in this 25.0 mL solution? Add 5.0 mL of the 25.0 mL Fe3+ solution to test tube #3. Remember that the tube already contains KSCN. What is [Fe3+]o for test tube #3? What is [SCN- ]o for test tube #3? Test Tube #4 Again, discard all but 10.0 mL of the Fe3+ solution in the graduated cylinder.
To this 10.0 mL, add water to get 25.0 mL of solution. Mix thoroughly. What is [Fe3+] in this 25.0 mL solution? Add 5.0 mL of the 25.0 mL Fe3+ solution to test tube #4. Remember that the tube already contains KSCN. What is [Fe3+]o for test tube #4? What is [SCN- ]o for test tube #4?