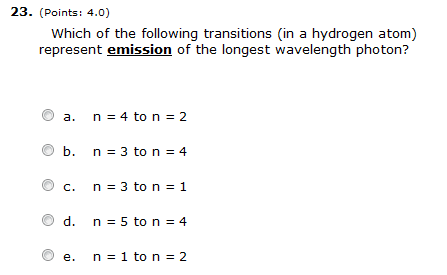9
answers
0
watching
314
views
23 Aug 2018
3. Which of the following electron transitions corresponds to emission of the longest wavelength light?
A. from n = 3 to n = 2
B. from n = 1 to n = 5
C. from n = 4 to n = 2
D. from n = 1 to n = 2
E. from n = 5 to n = 1
4. Emission from an excited Hg atom emits at 254 nm. What is the frequency of this light?
A: 1.18e15 sec-1
B: 1.18e15 sec
C: 1.18e6 sec-1
D: 1.18e6 sec
3. Which of the following electron transitions corresponds to emission of the longest wavelength light?
A. from n = 3 to n = 2
B. from n = 1 to n = 5
C. from n = 4 to n = 2
D. from n = 1 to n = 2
E. from n = 5 to n = 1
4. Emission from an excited Hg atom emits at 254 nm. What is the frequency of this light?
A: 1.18e15 sec-1
B: 1.18e15 sec
C: 1.18e6 sec-1
D: 1.18e6 sec
lalithashwin156Lv10
30 May 2023
akunuru639Lv10
28 May 2023
Already have an account? Log in
hardiksahuLv10
17 May 2023
Already have an account? Log in
2 Jun 2021
Already have an account? Log in