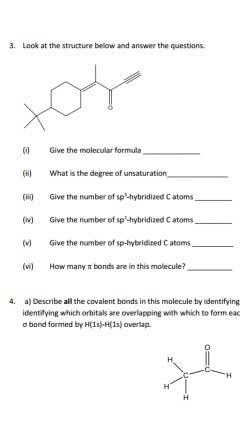
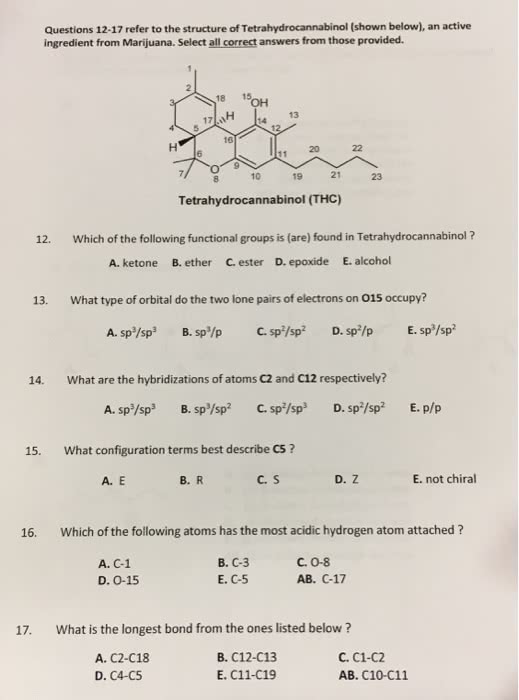
For questions 1-5 choose all correct answers that apply: Which of the following atoms has an electric configuration of 1s' 2s2 2p? E. Fluorine A. Boron B. Carbon 2. What is the molecular shape and the hybridization of the carbon atom at position number 2 in hex-3-yn-2-one? B. Tetrahedral, sp C. Trigonal planar, sp A. Tetrahedral, sp D. Bent, sp E. Linear, sp AB. Trigonal pyramidal, sp 3. Which molecule(s) is/are a constitutional (structural) isomer(s) of cyclohexanol ? A. 2-Hexanone B. 2-Methylcyclopentanone C. 1-Methoxypentane D. Hexanoic acid E. Hexanal 4. Which of the following isomers will have the most exothermic heat of formation: B. C. D. E. 5. Which following molecule(s) has/have the S-stereochemistry? A. B. C. D. E.