CHEM 113 Lecture Notes - Buffer Solution
51 views1 pages
Document Summary
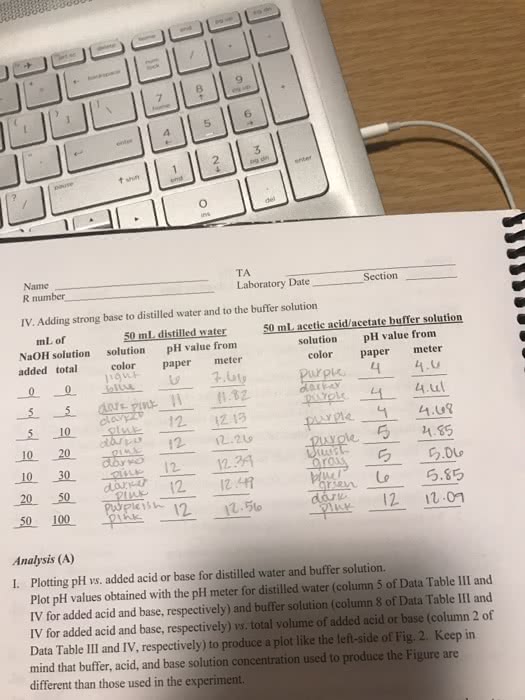
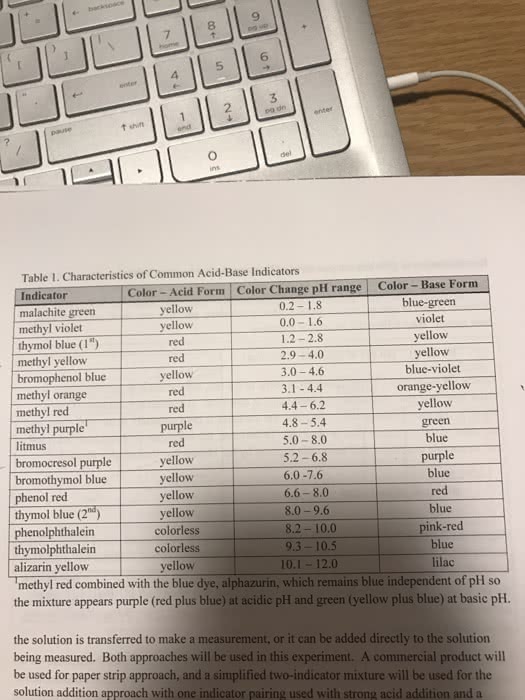
Because ph = pka + log [a+] / [ha], start with ph = pka. Then, determine [ha] and [a-] to give you the exact ph. Buffer capacity the ability of the buffer solution to resist changes in ph. More concentrated buffer components = greater buffer capacity. Buffer range the ph range over which the buffer acts effectively. [a-] / [ha] should be close to 1 (no larger than 10, no smaller than 0. 1) This will give you an effective ph range of +/- 1. The farther this ratio gets from 1, the less efficient the buffer (the smaller the buffer range)
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232