CHEM 212 Chapter Notes -Sodium Benzoate, Solvent, Benzoic Acid
Document Summary
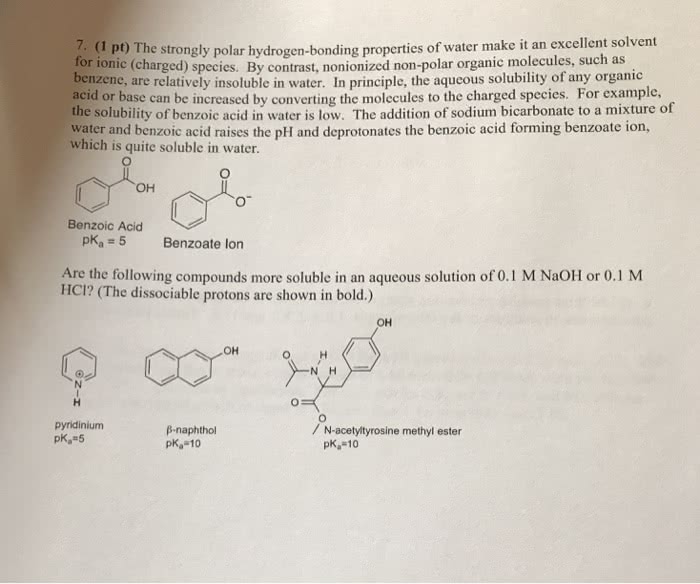
(a) benzoic acid itself in water is insoluble due to the hydrophobic benzene ring which disrupt the h-bonds between water-water and water-benzoic acid. It is more soluble i ether as it shares same properties with ether in molecular terms. Since it is an ionic solid, therefore, it is soluble in water since due to polar properties of water, all polar and ionic solids dissolve in water. (c) 1. Ether has lower density than water which means that top layer will be ether and bottom layer will be water. This means that benzoic acid will be present in top layer: since on mixing with naoh, benzoic acid forms sodium benzoate which is more soluble in water, therefore, benzoic acid will be in 3m naoh layer. Since, ccl4 has more density than water, therefore, ccl4 will be bottom later and 3m naoh will be top layer.