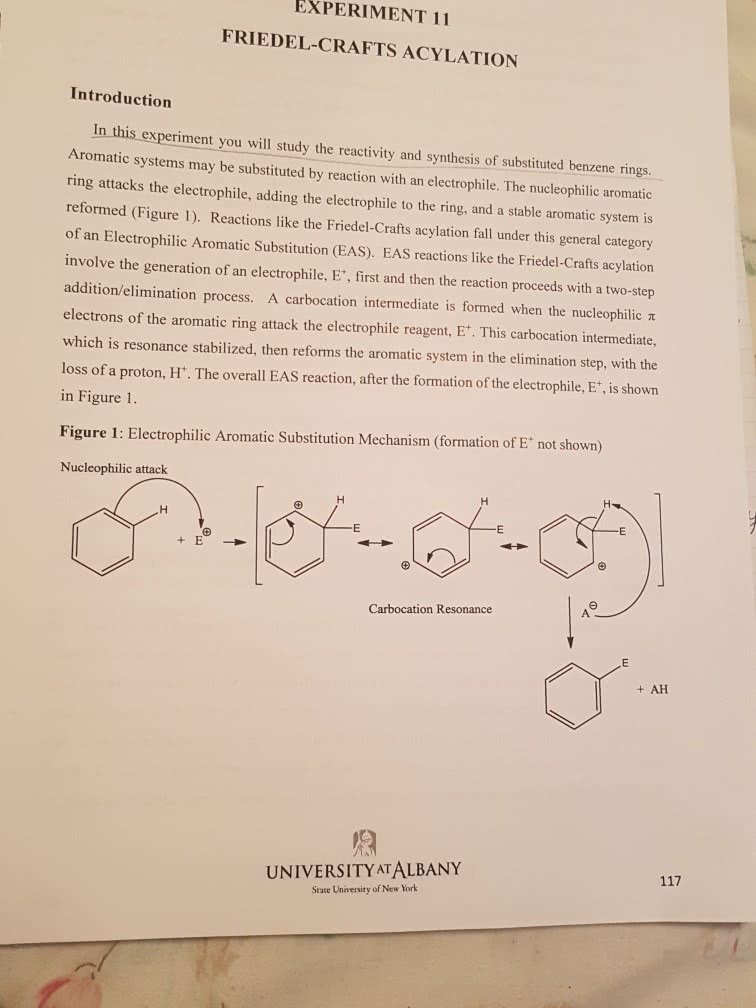
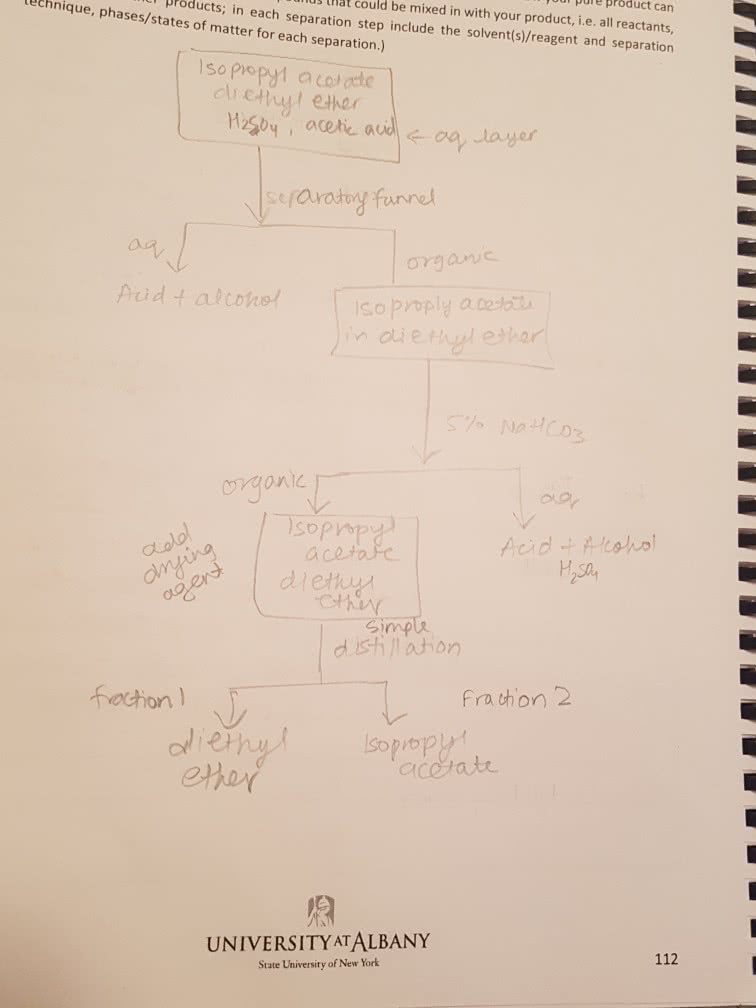
CHM136H1 : Notes about different reactions required for term 2

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
Generally, cis is less stable than trans, due to steric strain between the substituted chains (r group). Less stable compounds have less g0 and h0 values. The more substituted the compound is, the more stable the compound, and the greater their. Stability in alkenes are attributed by two factors: hyperconjugation. The interaction of a paired pi orbital and a neighbouring filled c h sigma bond on a substituent: bond strength. A sp2 sp3 carbon bond is stronger than a sp3 sp3 carbon bond. ** therefore, the anions would usually making bond with more substituted carbon, whereas the. Stability of carbocations (which is formed in step 4 of the above reaction mechanism: similar to the stability of alkenes, the more substituted it is, the more stable it is. Because with more r group around that carbocation, more inductive effect takes place.