CHEM 212 Chapter Notes - Chapter 1: Nail Polish, Propylene Oxide, Formal Charge
Document Summary
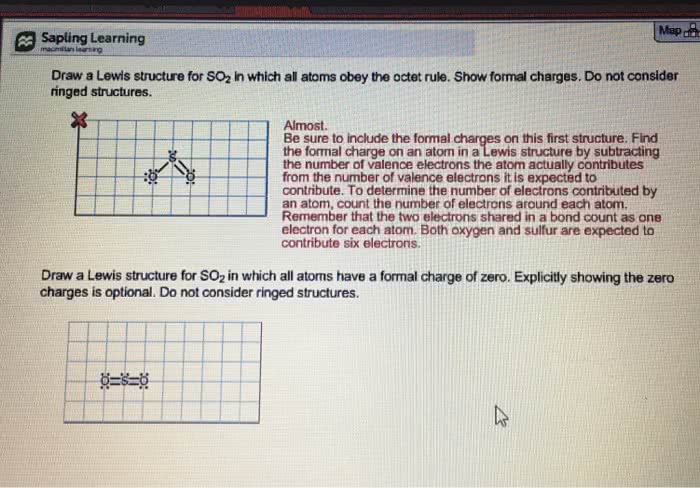
Many lewis structures are incomplete until we decide whether any of their atoms have a formal charge. Calculating the formal charge on an atom in a lewis structure is simply a bookkeeping method for its valence electrons. F irst, we examine each atom and, using the periodic table, we determine how many valence electrons it would have if it were an atom not bonded to any other atoms. This is equal to the group number of the atom in the periodic table. For hydrogen this number equals 1, for carbon it equals 4, for nitrogen it equals 5, and for oxygen it equals 6. Next, we examine the atom in the lewis structure and we assign the valence electrons in the following way: We assign to each atom half of the electrons it is sharing with another atom and all of its unshared (lone) electron pairs. Then we do the following calculation for the atom: