CHY 103 Chapter Notes - Chapter 16: Sodium Chloride, Phenolphthalein, Titration Curve
Document Summary
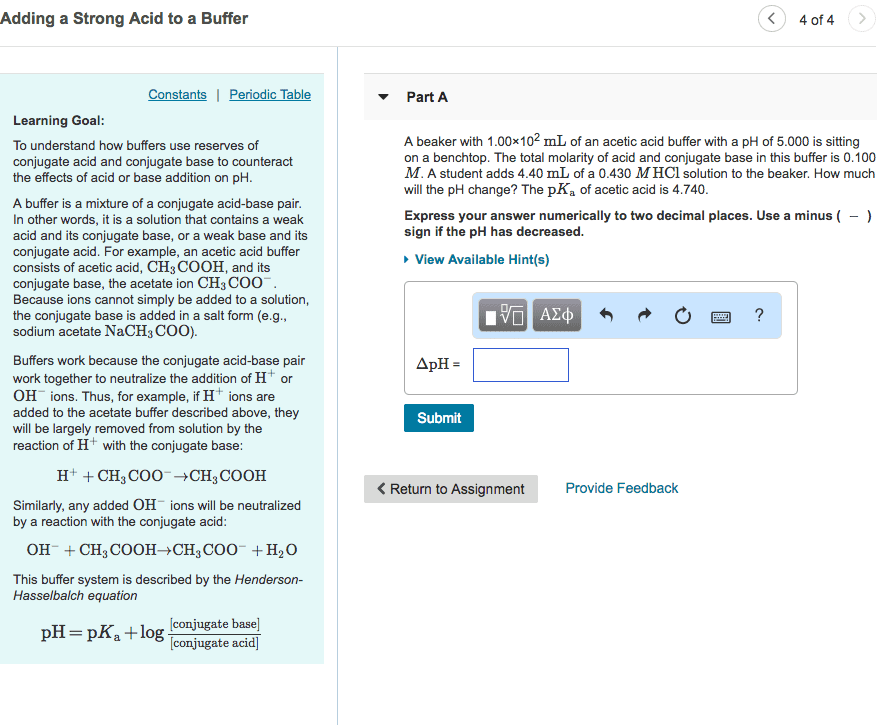
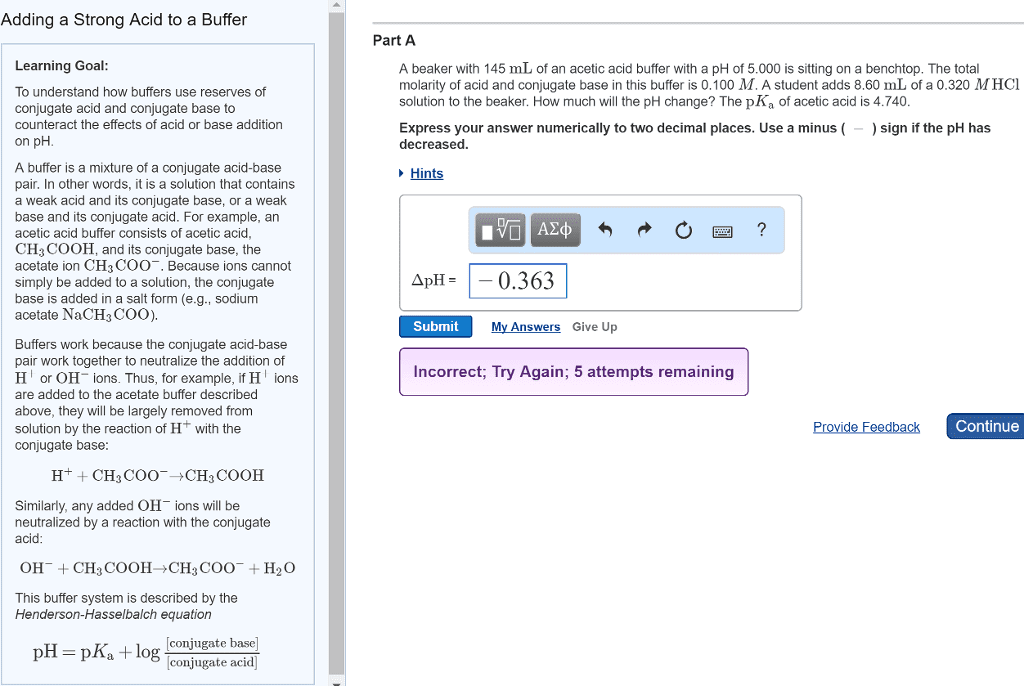
In this way, a buffer can maintain a nearly constant ph: a buffer must contain significant amounts of both a weak acid and its conjugate base. Consider the simple buffer made by dissolving acetic acid (ch3cooh) and sodium acetate (ch3coona) in water. Suppose that we add a strong bas, such as naoh to this solution. Acetic acid reacts with the oh- from naoh, neutralizing the base according to the following net ionic equation: (cid:1841)(cid:1834) (cid:4666)(cid:1853)(cid:1869)(cid:4667)+(cid:1834)(cid:2871)(cid:1841)(cid:1841)(cid:1834)(cid:4666)(cid:1853)(cid:1869)(cid:4667) (cid:1834)(cid:2870)(cid:1841)(cid:4666)(cid:1864)(cid:4667)+(cid:1834)(cid:2871)(cid:1841)(cid:1841) (cid:4666)(cid:1853)(cid:1869)(cid:4667) As long as the amount of added naoh is less than the amount of (cid:1834)(cid:2871)(cid:1841)(cid:1841)(cid:1834) in solution, the buffer neutralizes the added naoh and the resulting ph change is small. Summary of a buffer: buffers resist ph change, a buffer contains significant amounts of both a weak acid and its conjugate base, the weak acid neutralizes added base, the conjugate base neutralizes added acid.