10
answers
0
watching
783
views
29 Mar 2020
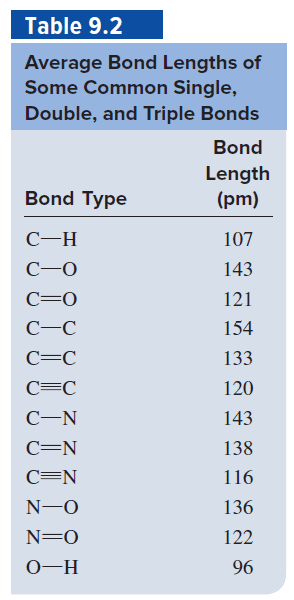
The bond lengths of carbon–carbon, carbon–nitrogen, carbon–oxygen, and nitrogen–nitrogen single, double, and triple bonds are listed in Table 8.5. Plot bond enthalpy (Table 8.4) versus bond length for these bonds (as in Figure 8.17). (a) Is this statement true: “The longer the bond, the stronger the bond”? (b) Order the relative strengths of C – C, C – N, C – O, and N – N bonds from weakest to strongest. (c) From your graph for the carbon–carbon bond in part (a), estimate the bond enthalpy of the hypothetical CC quadruple bond.
The bond lengths of carbon–carbon, carbon–nitrogen, carbon–oxygen, and nitrogen–nitrogen single, double, and triple bonds are listed in Table 8.5. Plot bond enthalpy (Table 8.4) versus bond length for these bonds (as in Figure 8.17). (a) Is this statement true: “The longer the bond, the stronger the bond”? (b) Order the relative strengths of C – C, C – N, C – O, and N – N bonds from weakest to strongest. (c) From your graph for the carbon–carbon bond in part (a), estimate the bond enthalpy of the hypothetical CC quadruple bond.
marcusnicole284Lv10
2 Jul 2023
Already have an account? Log in
3 Mar 2023
Already have an account? Log in
malupiton2022Lv10
12 Oct 2022
Already have an account? Log in
papayaprofessorLv10
5 Sep 2022
Already have an account? Log in
Casey DurganLv2
21 May 2020
Already have an account? Log in