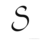16
answers
0
watching
418
views
26 Sep 2019
Below are the statements about entropy. Identify the correct statement.
-
There is no change in entropy for a reversible process
-
Entropy for an irreversible process always decreases
-
The unit of entropy is KJ/mol
-
Entropy of an isolated system always decreases
Below are the statements about entropy. Identify the correct statement.
-
There is no change in entropy for a reversible process
-
Entropy for an irreversible process always decreases
-
The unit of entropy is KJ/mol
-
Entropy of an isolated system always decreases
teacherrecoLv10
6 Aug 2023
devsingh077Lv10
16 Jul 2023
Already have an account? Log in
17 Jun 2023
Already have an account? Log in
viclere2005Lv10
8 May 2023
Already have an account? Log in
24 Dec 2021
Already have an account? Log in
madhusaranithaaLv10
27 Oct 2021
Already have an account? Log in
Keith LeannonLv2
1 Feb 2019
Already have an account? Log in