1. The most useful way torationalize the strengths of uncharged acids is to compare thestabilities of the conjugate bases (negatively charged) formed byeach acid. The strongest acid will generate the most stableconjugate base, while the weakest acid will lead to formation ofthe least stable conjugate base. (Or you can say that the strongestacid yields the weakest conjugate base, and the weakest acid yieldsthe strongest conjugate base.) Use this strategy to explain thefollowing pKa values.
a. Alcohols are strongeracids than amines, and amines are stronger acids than alkanes.Explain. (Three examples are shown below.)
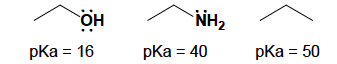
b. Acetic acid,CH3CO2H, is astronger acid than ethanol, CH3CH2OH (pKa = 5 and 16,respectively). Why?
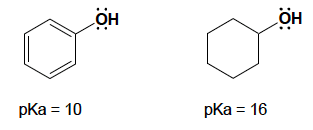
c. Phenol is a stronger acidthan cyclohexanol. Explain.
The most useful way to rationalize the strengths of uncharged acids is to compare the stabilities of the conjugate bases (negatively charged) formed by each acid. The strongest acid will generate the most stable conjugate base, while the weakest acid will lead to formation of the least stable conjugate base. (Or you can say that the strongest acid yields the weakest conjugate base, and the weakest acid yields the strongest conjugate base.) Use this strategy to explain the following pKa values. Alcohols are stronger acids than amines, and amines are stronger acids than alkanes. Explain. (Three examples are shown below.) Acetic acid, CH3CO2H, is a stronger acid than ethanol, CH3CH2OH (pKa = 5 and 16, respectively). Why?