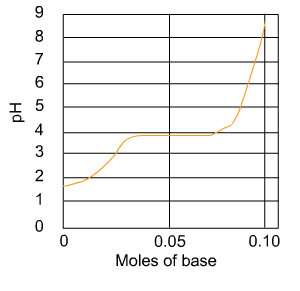
 100mlof 1.0 M formic acid (HCOOH) is titrated with 1.0M sodium
100mlof 1.0 M formic acid (HCOOH) is titrated with 1.0M sodium
hydroxide (NaOH). The titration curve is shown.
a.) What is the apporximate Pka?
b.) At the pKa what fraction of the carboxyl group willhave
been coverted to COO-?
formic acid (HCOOH) is titrated with 1.0M sodium hydroxide (NaOH). The titration curve is shown. What is the approximate Pka? At the pKa what fraction of the carboxyl group will have been converted to COO-?
Show transcribed image text formic acid (HCOOH) is titrated with 1.0M sodium hydroxide (NaOH). The titration curve is shown. What is the approximate Pka? At the pKa what fraction of the carboxyl group will have been converted to COO-?